stgitskaysie9028
22.02.2021 •
Chemistry
Identify the number of valence electrons in each atom:
a. Calcium
b. Carbon
c. Sulfur
Solved
Show answers
More tips
- L Leisure and Entertainment What Movies You Should Watch: A Guideline to Make the Right Decision...
- F Family and Home How to Choose a Baby Stroller: Tips from Experienced Parents...
- H Health and Medicine 5 Simple Steps to Quit Smoking for Good...
- C Computers and Internet How to Download Videos from YouTube? Simple Steps to Download Any Content...
- H Health and Medicine What is the Normal Blood Sugar Level in a Healthy Person?...
- S Style and Beauty How to Get Rid of Acne: Scientifically Proven Methods...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- C Computers and Internet How to Delete Your Account on Odnoklassniki...
- H Health and Medicine 10 Simple and Effective Tips on How to Lose Weight in a Week...
- F Food and Cooking What s the Best Rice for Cooking Plov?...
Answers on questions: Chemistry
- C Chemistry Joey was taking a chemistry test. in one question, he was asked to write the electron configuration for antimony (sb). he wrote it out like this: 1s22s22p63s23p63d104s24p64d105s25p3...
- C Chemistry Notice that s o 4 appears in two different places in this chemical equation. s o 2− 4 is a polyatomic ion called sulfate. what number should be placed in front of cas...
- C Chemistry One mole of air at 310 k confined in a cylinder under a heavy piston occupies a volume of 4.9 l. the heat capacity of air under constant volume is 5 2 r. find the new...
- C Chemistry The energy stored in the bonds between atoms is a. chemical energy b. thermal energy c. electrical energy d. endothermic energy...
- C Chemistry Rubbing alcohol is 70.0% isopropyl alcohol by volume. how many ml of isopropyl alcohol are in a full 1 pint (473 ml) container?...
- C Chemistry Iwill reward this is science...
- C Chemistry Suppose a solution was too concentrated for an accurate reading with the spectrophotometer. the concentrated solution was diluted by placing 1.00 ml of the concentrated...
- C Chemistry Uranium-235 and plutonium-239 are used in nuclear reactors because of their 1. ability to undergo fission 2. ability to undergo fusion 3. inability to absorb neutrons...
- C Chemistry What is the gram formula mass of glycine , nh2ch2cooh...
- C Chemistry An engineer that designs buildings is a (n)...

Ответ:
Calcium - 2
Carbon - 4
Sulfur- 6
Ответ:
-Sulfur is a chemical element with the symbol S and atomic number 16. It is abundant, multivalent and nonmetallic. Under normal conditions, sulfur atoms form cyclic octatomic molecules with a chemical formula S₈. Elemental sulfur is a bright yellow, crystalline solid at room temperature.
(Atomic number: 16
Electron configuration: [Ne] 3s²3p⁴
Electrons per shell: 2,8,6
Electronegativity: 2.58)
-Calcium is the 20th element, with 20 protons. Since a stable atom has a net charge of 0, we must have 20 electrons.
-Carbon is a chemical element with the symbol C and atomic number 6. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. It belongs to group 14 of the periodic table. Carbon makes up only about 0.025 percent of Earth's crust.
(Symbol: C
Atomic mass: 12.0107 u
Atomic number: 6
Electrons per shell: 2, 4
Electron configuration: [He] 2s22p2
Electronegativity: 2.55)
Ответ:
The mass of aluminium hydroxide needed is 150.774 grams.
Explanation:
The chemical equation for the decomposition of aluminium hydroxide follows:
At STP:
22.4 L of volume is occupied by 1 mole of a gas.
So, 65 L of volume will be occupied by =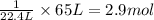
By Stoichiometry of the reaction:
3 moles of water is produced by 2 moles of aluminium hydroxide
So, 2.9 moles of water is produced by =
To calculate the mass of aluminium hydroxide, we use the equation:
Number of moles of aluminium hydroxide = 1.933 moles
Molar mass of aluminium hydroxide = 78 g/mol
Putting values in above equation, we get:
Hence, the mass of aluminium hydroxide needed is 150.774 grams.