Identify the spectator ions in this reaction. check all that apply. 2h+ + cro4– + ba2+ + 2oh– ba2+ + cro4– + 2h2o h+ cro4– ba2+ oh– 2h2o
Solved
Show answers
More tips
- S Style and Beauty Don t Sacrifice Your Brows: How to Properly Pluck Stubborn Hairs...
- W Work and Career Secrets of a Super Supervisor: Proper Team Management...
- P Photography and Videography Understanding HDR: How It Works and Why You Need It...
- G Goods and services Which TV is better - LCD or Plasma?...
- S Sport How to Learn to Pull Up on Monkey Bars?...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- C Computers and Internet Where did torrents.ru move to?...
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
Answers on questions: Chemistry
- C Chemistry The quantity of heat required to raise the temperature of 1 gram of sample of a substance by 1°C is the sample’s Question 2 options: heat capacity. specific heat capacity....
- C Chemistry The acid dissociation constant of alloxanic acid is . Calculate the pH of a solution of alloxanic acid. Round your answer to decimal place....
- C Chemistry Testbank Question 54 Dissolving benzaldehyde in methyl alcohol establishes an equilibrium with what compound?...
- C Chemistry clacium hydroxide is slightly soluable in water about 1 gram will dissolve in 1 liter what are the spectator ions in the reaction ions in the reaction of such a dilute...
- C Chemistry Achemist is performing a titration in order to determine the amount of lithium hydroxide, lioh(aq), in 135 ml of an aqueous solution. (a) if 28 ml of a 0.15 m hcl solution...
- C Chemistry Adna molecule is said to unzip when it is involved with cell division, which means the two strands of the molecule separate in order to combine with new strands. which...
- C Chemistry Using the following equation, how many moles of O2 will be formed when 1.65 moles of KClO3 decomposes?...
- C Chemistry a person tries to catch a bus to see how it starts; in a desperate attempt, he starts running with a speed of 6m / s. When you are 32 m from the bus stop, it starts with...
- C Chemistry Reaction is found to have a rate constant of 12.5 s-1 at 25.0 Celsius. When you heat the reaction up by ten degrees Celsius, the rate of the reaction exactly doubles....
- C Chemistry A gas contained in a steel tank has a pressure of 1.5 atm at a temperature of 320 K. What will be the gas pressure when the temperature changes to 450 K?...

Ответ:
Explanation:
1) Chemical equation given:
2H⁺ + CrO₄⁻ + Ba²⁺ + 2OH⁻ → Ba²⁺ + CrO₄⁻ + 2H₂O
2) Analysis
That is an oxidation-reduction equation (some species are been oxidized and others are being reduced).
The given equation is known as total ionic equation, because it shows all the species as ions that are part of the reaction.
2) Specator ions
Spectator ions are the ions that do not change their oxidation state and are easily identified as they are the same in the reactant and product sides.
Here the ions that are the same in the reactant and product sides are:
CrO₄⁻ and Ba²⁺
3) Addtitional explanation.
Once you identify the spectator ions you can delete them from the equation to obtain the net ionic equation , which in this case turns to be:
2H⁺ + 2OH⁻ → 2H₂O
But this is not part of the question; it is some context to help you understand the use of the spectator ions concept.
Ответ:
The question is incomplete, the complete question is:
A 7.00 L tank at is filled with 5.43 g of sulfur hexafluoride gas and 14.2 g of sulfur tetrafluoride gas. You can assume both gases behave as ideal gases under these conditions. Calculate the mole fraction and partial pressure of each gas. Round each of your answers to significant digits.
is filled with 5.43 g of sulfur hexafluoride gas and 14.2 g of sulfur tetrafluoride gas. You can assume both gases behave as ideal gases under these conditions. Calculate the mole fraction and partial pressure of each gas. Round each of your answers to significant digits.
The mole fraction of sulfur hexafluoride is 0.221 and that of sulfur tetrafluoride is 0.779
Explanation:
The number of moles is defined as the ratio of the mass of a substance to its molar mass. The equation used is:
 ......(1)
......(1)
For sulfur hexafluoride:Given mass of sulfur hexafluoride = 5.43 g
Molar mass of sulfur hexafluoride = 146.06 g/mol
Putting values in equation 1, we get:
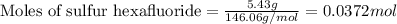
For sulfur tetrafluoride:Given mass of sulfur tetrafluoride = 14.2 g
Molar mass of sulfur tetrafluoride = 108.07 g/mol
Putting values in equation 1, we get:
Total moles of gas in the tank = [0.0372+ 0.1314] mol = 0.1686 mol
Mole fraction is defined as the moles of a component present in the total moles of a solution. It is given by the equation:
where n is the number of moles
Putting values in equation 2, we get:
Hence, the mole fraction of sulfur hexafluoride is 0.221 and that of sulfur tetrafluoride is 0.779