catelinboog04
21.11.2020 •
Chemistry
If 9.8g water is used in electrolysis, what is the percent yield if 5.6g of oxygen was
collected after the experiment?
H20 --> H2 + O2
Solved
Show answers
More tips
- P Photography and Videography Understanding HDR: How It Works and Why You Need It...
- G Goods and services Which TV is better - LCD or Plasma?...
- S Sport How to Learn to Pull Up on Monkey Bars?...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- C Computers and Internet Where did torrents.ru move to?...
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
- G Goods and services How to Choose the Right High Chair for Your Baby?...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
Answers on questions: Chemistry
- C Chemistry What does a chemical reaction always produce?A) A different phase of matter (solid from liquid, gas from liquid, etc.)B) A physical changeC) A chemical changeD)...
- C Chemistry How much heat must be transferred to 100.0g of water to change its temperature by 35° C?...
- C Chemistry Connections related to to science and engineering practices...
- C Chemistry Draw the structure of a skeletal muscle that releases a calcium ion in response to the arrival of the action potential....
- C Chemistry The ability of an atom in a molecule to attract electrons is best quantified by the . a. electron charge-to-mass ratio b. electronegativity c. first ionization potential...
- C Chemistry 21. Which statement about one atom of an element identifies the element? A) The atom has 1 proton. B) The atom has 2 neutrons. C) The sum of the number of protons...
- C Chemistry Marigold, Inc. uses activity-based costing as the basis for information to set prices for its six lines of seasonal coats. Activity Cost Pools Estimated Overhead...
- C Chemistry How do lone electron pairs affect molecular shape? Please help! I know it has to do with the VSEPR theory, but I don t know how, so please explain the best you can!...
- C Chemistry Label the layers of the atmosphere in the diagram below. help me!!...
- C Chemistry Good morning beautiful people of the world hope yall have a wonderful day Follow me on instagram maybe branasia07...

Ответ:
Approximately .
.
Explanation:
The actual yield of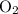 was given. The theoretical yield needs to be calculated from the quantity of the reactant.
was given. The theoretical yield needs to be calculated from the quantity of the reactant.
Balance the equation for the hydrolysis of water:
Note the ratio between the coefficient of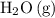 and
and  :
:
This ratio will be useful for finding the theoretical yield of .
.
Look up the relative atomic mass of hydrogen and oxygen on a modern periodic table.
Calculate the formula mass of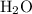 and
and 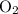 :
:
Calculate the number of moles of molecules in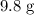 of
of 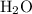 :
:
Make use of the ratio to find the theoretical yield of
to find the theoretical yield of 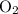 (in terms of number of moles of molecules.)
(in terms of number of moles of molecules.)
Calculate the mass of that approximately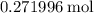 of
of 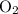 (theoretical yield.)
(theoretical yield.)
That would correspond to the theoretical yield of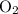 (in term of the mass of the product.)
(in term of the mass of the product.)
Given that the actual yield is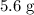 , calculate the percentage yield:
, calculate the percentage yield:
Ответ:
As we know, 1 mole of any substance contains 6.022 × 10²³ particles. So, 1 mole of Na₂SO₄ also contains 6.022 × 10²³ formula units of Na₂SO₄.
Also,
1 mole of Na₂SO₄ contains = 6.022 × 10²³ Atoms of S
So,
4.5 moles of Na₂SO₄ will contain = X atoms of S
Solving for X,
X = (4.5 moles × 6.022 × 10²³ Atoms) ÷ 1 mole
X = 2.70 × 10²⁴ Atoms of S
Result:
4.5 Moles of Na₂SO₄ contains 2.70 × 10²⁴ Atoms of Sulfur.