tankhill5534
09.04.2021 •
Chemistry
If I have a sealed tank of oxygen at 85 degrees celsius and I cool it down to 50 degrees celsius. What happens to the pressure? *
Solved
Show answers
More tips
- L Leisure and Entertainment Should You Buy a Ceramic Knife?...
- C Computers and Internet How to easily and quickly disable Firebug in Gmail and Google Docs...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
Answers on questions: Chemistry
- S Social Studies explain how cross-cultural interactions resulted in the d and facilitaed changes in patterns of trade and travel...
- A Arts For one to be able to write an song what is the first step in the song writing process?...
- H History What is the difference between inflation and deflation? Inflation can result from falling demand and boosts the value of money. Deflation can result from rising...
- M Mathematics Y=8x+7 i need helpppp...

Ответ:
Answer : The correct option is (d) a solution of 0.10 M NaOH
Explanation :
(a) a solution of pH 3.0
First we have to calculate the pOH.
Now we have to calculate the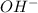 concentration.
concentration.
Thus, the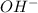 concentration is,
concentration is, 
(b) a solution of 0.10 M
As we know that 1 mole of is a weak base. So, in a solution it will not dissociates completely.
is a weak base. So, in a solution it will not dissociates completely.
So, the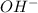 concentration will be less than 0.10 M
concentration will be less than 0.10 M
(c) a solution with a pOH of 12.
We have to calculate the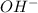 concentration.
concentration.
Thus, the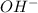 concentration is,
concentration is, 
(d) a solution of 0.10 M NaOH
As we know that is a strong base. So, it dissociates to give
is a strong base. So, it dissociates to give 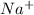 ion and
ion and 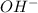 ion.
ion.
So, 0.10 M of in a solution dissociates to give 0.10 M of
in a solution dissociates to give 0.10 M of 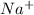 ion and 0.10 M of
ion and 0.10 M of 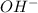 ion.
ion.
Thus, the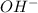 concentration is, 0.10 M
concentration is, 0.10 M
(e) a solution of
solution of 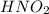
As we know that 1 mole of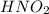 in a solution dissociates to give 1 mole of
in a solution dissociates to give 1 mole of  ion and 1 mole of
ion and 1 mole of 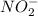 ion.
ion.
So, of
of 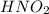 in a solution dissociates to give
in a solution dissociates to give  of
of  ion and
ion and  of
of 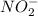 ion.
ion.
The concentration of ion is
ion is 
First we have to calculate the pH.
Now we have to calculate the pOH.
Now we have to calculate the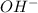 concentration.
concentration.
Thus, the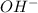 concentration is,
concentration is, 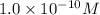
From this we conclude that, a solution of 0.10 M NaOH possess the greatest concentration of hydroxide ions.
Hence, the correct option is (d)