mricee9718
05.05.2020 •
Chemistry
If the pka of hcho2 is 3.74 and the ph of an hcho2/nacho2 solution is 3.89, it means that
Solved
Show answers
More tips
- D Dating, Love, Relationships Does a Person s Character Depend on the Color of Their Eyes?...
- O Other Childhood Fears: What Many of Us Experienced...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- L Leisure and Entertainment What to Bring on a Hike? Essential Items to Pack for a Safe and Enjoyable Adventure...
- L Leisure and Entertainment Couchsurfing: A New Way to Travel...
- S Style and Beauty Autotanning: Harmful or Safe?...
- F Food and Cooking 10 Ideas for a Wedding Anniversary Gift...
- H Health and Medicine How to Reduce Sweating in the Heat and Beyond: Say Goodbye to Excessive Sweat...
- F Food and Cooking Do Aphrodisiacs Really Work? Separating Fact from Fiction...
- H Health and Medicine What to Eat to Lose Weight?...
Answers on questions: Chemistry
- E English 2points what is one possible reason that kafka s the metamorphosis is still interesting to readers nearly 100 years after it was written? o a. the story represents the idea that...
- B Biology Select the correct answer. Why does soil become less fertile the deeper into the Earth it gets? OA The farther down, the more it is removed from organic matter. O B. The percentage...
- S SAT if p and q are non-zero rational numbers, and s and t are irrational numbers, which statement is possibly true?...
- C Chemistry What is the formula for manganese (IV) oxide? (5 points) MnO4 Mn4O MnO2 Mn2O...
- C Computers and Technology How to call a method inside of a react functional class component test....

Ответ:
Hi, the question has some missing part. The complete question should be " If the of
of 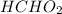 is 3.74 and the pH of an
is 3.74 and the pH of an  solution is 3.89, it means that -
solution is 3.89, it means that -
(a)![[HCHO_{2}]=[NaCHO_{2}]](/tpl/images/0643/4545/85113.png)
(b)![[HCHO_{2}] [NaCHO_{2}]](/tpl/images/0643/4545/e1490.png)
(c)![[HCHO_{2}]< [NaCHO_{2}]](/tpl/images/0643/4545/97796.png)
Which of the options are true?"
Option (c) is true.
Explanation:
Here, pH = 3.89, of
of 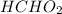 = 3.74
= 3.74
So,![3.89=3.74+log(\frac{[CHO_{2}^{-}]}{[HCHO_{2}]})](/tpl/images/0643/4545/d55a8.png)
or,![\frac{[CHO_{2}^{-}]}{[HCHO_{2}]}](/tpl/images/0643/4545/5aa24.png) = 1.41 (>1)
= 1.41 (>1)
So,![[CHO_{2}^{-}] [HCHO_{2}]](/tpl/images/0643/4545/877d3.png)
or,![[NaCHO_{2}] [HCHO_{2}]](/tpl/images/0643/4545/6ef0b.png)
Hence, option (c) is correct.
Ответ:
Cl
Explanation:
Because the oxidation state reduces from+3 in ClO2- to +1 in ClO- and oxygen is being lost