drippyc334
11.07.2019 •
Chemistry
If the same quantity of heat is added to both a 2-liter and a 4-liter container of water, the temperature change for water in the 4-liter container will be a.half that of the 2-liter container. b.more than half but less than twice. c.twice that of the 2-liter container. d.no change. explain.
Solved
Show answers
More tips
- A Art and Culture The History and Characteristics of Jazz Bands: A Deep Dive...
- F Food and Cooking From Latte to Espresso: Which Coffee Drink is the Most Popular on Earth?...
- C Computers and Internet How to Set Up Internet on iPhone? Detailed Guide with Step-by-Step Instructions...
- P Philosophy 8 привычек, чтобы достичь счастливой жизни...
- F Food and Cooking How Many Grams Are In a Tablespoon?...
- G Goods and services How to Choose the Right Iron: Purchase Tips...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- H Health and Medicine How to Choose the Right Glasses?...
- H Health and Medicine What vaccines do children need?...
- H Health and Medicine AKDS Vaccination: Ensure Your Child s Safety...
Answers on questions: Chemistry
- C Chemistry Select true if the sentences uses commas correctly, and select false if they are not used correctly. my english class, one of my favorite courses, takes place every morning true...
- C Chemistry Okay so this is for science and we have to have it done by tomorrow so can someone me with it we have to get the things for the blue bubbles...
- C Chemistry The reaction of a lewis acid with a lewis base would most likely br accomplished by an increase in which itema. couldn t bondsb. hydronium ionsc. electron pairsd. hydroxide ions...
- C Chemistry How does H3O+ and OH- relate to acids, bases, and the pH scale?...
- C Chemistry Which represents the balanced double displacement reaction between sodium chloride (na+cl-) and copper sulfate (cu2+(so4)2-) ? nacl + cu(so4) -- na2(so4) + cucl2 nacl + cu(so4)...
- C Chemistry Write the formulas for the ionic compounds formed by the following: 1. potassium & iodine 2. aluminum & bromine 3. cesium & nitrate 4. aluminum & carbonate...
- C Chemistry What is the relationship between lattice energy and bond strength? a. the higher the lattice energy, the stronger the bond b. the higher the lattice energy, the weaker the bond...
- C Chemistry Explain how an ionic compound forms between na & n. na atom(s) each lose electron(s), ion(s). atom(s) gains (s), forming a ion. the ions attract, forming na3n...
- C Chemistry Why are negative ions larger than the neutral atom from which they form? a. because the addition of electrons results in an added energy level b. because the addition of electrons...
- C Chemistry Ineed ! i’m torn between a, b and ! apex...

Ответ:
-
The temperature change for water in the 4-liter container will be
a.half that of the 2-liter container.
Explanation: -
The increase in temperature of a substance when heat is given to it is given by the formula
Q = m x c x ΔT
Where Q is the heat supplied, m is the mass of water, c is the specific heat of water and ΔT is the change in the temperature of water.
Or ΔT =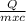
From the formula we see that for the same amount of heat, increase is temperature is more for less amount of mass.
We know that more the volume of water, more the amount of water present and consequently more the mass of water.
Thus for the same amount of heat supplied, the temperature increases more for a lesser volume of water.
Let density of water be d.
Mass of 4 L= volume x density = 4 L x d
T 1 = ΔT for 4 L =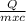
=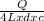
Mass of 2 L= volume x density = 2 L x d
T2 = ΔT for 2 L =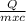
=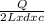
On taking ratio
Thus the temperature change for water in the 4-liter container will be
a.half that of the 2-liter container.
Ответ: