myapawesomeox22sq
03.02.2021 •
Chemistry
If you cooled 85g of water from 55c° to 2c°, what is the amount of heat lost
Solved
Show answers
More tips
- H Health and Medicine Everything You Wanted to Know About Male Circumcision: Its Goals and Effects...
- L Leisure and Entertainment Couchsurfing: A New Way to Travel...
- G Goods and services Which TV is better - LCD or Plasma?...
- S Sport How to Learn to Pull Up on Monkey Bars?...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- C Computers and Internet Where did torrents.ru move to?...
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
- G Goods and services How to Choose the Right High Chair for Your Baby?...
Answers on questions: Chemistry
- C Chemistry Conservation Activity What roles do society, politics, and economics play in science? Discovering the roles and influence that society, politics, and economics have on science takes...
- M Mathematics A peice of wire is 240cm long. Peter cuts two 45cm lengths off the wire. He then cuts the rest of the wire into as many 40cm lengths as possible. Work out how many 40cm lengths of...
- B Biology Scientist have discovered a specific mineral that is radioactive and can be traced as it changed forms. One scientist wonders if they traced the mineral long enough if it would become...
- E English Which statement is an effective claim? (10 points) a I think mini-vans are more practical than economy cars. b Rules about teenage curfews should be changed. c There are several advantages...
- H Health The most important aspect of successful psychotherapy is the between client and therapist....

Ответ:
Q = - 18830.9 J
Explanation:
Given data:
Mass of water = 85 g
Initial temperature = 55°C
Final temperature = 2°C
Amount of heat lost = ?
Solution:
Specific heat capacity:
It is the amount of heat required to raise the temperature of one gram of substance by one degree.
Specific heat capacity of water 4.18 j/g.°C
Formula:
Q = m.c. ΔT
Q = amount of heat absorbed or released
m = mass of given substance
c = specific heat capacity of substance
ΔT = change in temperature
ΔT = 2°C - 55°C
ΔT = -53°C
Q = 85 g × 4.18 j/g.°C × -53°C
Q = - 18830.9 J
Negative sign shows that heat is released.
Ответ:
1) La masa del agua a temperatura ambiente es de 500 gramos, 2) La masa del agua cuando se congela es de 500 gramos, 3) La masa de agua que queda después de la evaporación es de 400 gramos, 4) Se ha evaporado 100 gramos de agua.
Explanation:
1) ¿Cuál es la masa de agua a temperatura ambiente?
Podemos determinar la masa inicial del agua (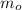 ), medido en gramos, al conocer su densidad (
), medido en gramos, al conocer su densidad (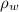 ), medida en gramos por mililitro, y volumen inicial ocupado en el vaso de precipitado (
), medida en gramos por mililitro, y volumen inicial ocupado en el vaso de precipitado ( ), medido en mililitros, a partir de la siguiente expresión:
), medido en mililitros, a partir de la siguiente expresión:
Si sabemos que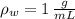 y
y  , entonces:
, entonces:
La masa del agua a temperatura ambiente es de 500 gramos.
2) ¿Cuál es la masa de agua cuando se congela?
Puesto que el proceso de congelación no implica transferencia de masa, la masa de agua se conserva al transformarse en hielo. Por tanto, la masa resultante es de 500 gramos.
3) ¿Cuál es la masa de agua que queda después de la evaporación?
Durante la evaporación una parte del agua es transferida al aire, entonces podemos calcular la masa final (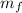 ), medido en gramos, de la sustancia al multiplicar el volumen final (
), medido en gramos, de la sustancia al multiplicar el volumen final (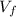 ), medido en mililitros, por la densidad del agua (
), medido en mililitros, por la densidad del agua (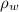 ), medida en gramos por mililitro,. Es decir,
), medida en gramos por mililitro,. Es decir,
Si sabemos que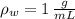 y
y 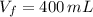 , entonces:
, entonces:
La masa de agua que queda después de la evaporación es de 400 gramos.
4) ¿Qué masa de agua se evaporó?
Determinamos que la masa evaporada de agua (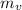 ), medida en gramos, es igual a la diferencia entre las masas inicial y final, ambas medidas en gramos:
), medida en gramos, es igual a la diferencia entre las masas inicial y final, ambas medidas en gramos:
Si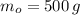 y
y 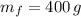 , entonces tenemos que:
, entonces tenemos que:
Se ha evaporado 100 gramos de agua.