mikemurray115
11.03.2020 •
Chemistry
If you have 0.82 moles of an ideal gas at 49k and a volume of 5.6L what would be the pressure
Solved
Show answers
More tips
- T Travel and tourism Lost in the Catacombs: What to Do?...
- L Leisure and Entertainment What to Bring on a Hike? Essential Items to Pack for a Safe and Enjoyable Adventure...
- S Style and Beauty How Are Eyelash Extensions Applied? All Your Questions Answered...
- F Family and Home Tender Care for Your Parquet: Is it Possible to Clean Parquet?...
- S Society and Politics Is It Fact or Fiction? Let s Talk About Anton Chekhov s Pseudonym...
- S Sport Playing Bowling: Rules and Advice for Novices...
- C Computers and Internet How to Properly Repartition a Hard Drive?...
- A Auto and Moto What Is the Cost of Customs Clearance for a Car in Russia?...
- L Leisure and Entertainment Should You Buy a Ceramic Knife?...
- C Computers and Internet How to easily and quickly disable Firebug in Gmail and Google Docs...
Answers on questions: Chemistry
- C Chemistry If 1.20 moles of copper react with mercuric nitrate, how many moles of mercury form? Cu + Hg(NO3)2 Cu(NO3)2 + Hg I WILL GIVE Brainliest TO THE RIGHT ANSWER...
- E English Which of the following explanations best interprets the meaning of the following sentence? the church was not merely a thermometer that recorded the ideas and principles...
- M Mathematics Red ribbons are wrapped around all the columns of a building. each ribbon does not overlap itself. each column has a radius of 3 feet. if all the ribbons are taken off the...
- M Mathematics A bag of trail mix weighs 1.625 pounds. Round 1.625 to the nearest tenth....

Ответ:
The pressure of the gas is 0.590 atm.
Explanation:
Pressure of the gas = P
Volume of the gas = V = 5.6 L
Temperature of the gas = T = 49 K
Moles of the gas = 0.82 mole
Using ideal gas equation :
P = 0.590 atm
The pressure of the gas is 0.590 atm.
Ответ:
The degree of ionization of a weak acid is greater than 50% but less than 100%.
Explanation:
An acid which dissociates partially or weakly when dissolved in water is called a weak acid.
For example, acetic acid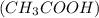 is a weak acid.
is a weak acid.
Hence, the degree of ionization of a weak acid is greater than 50% but less than 100%.
Thus, we can conclude that the degree of ionization of a weak acid is greater than 50% but less than 100%.