jahayla4life
01.09.2019 •
Chemistry
If you start with 4.00 moles of c3h8 (propane) and 4.00 moles of o2, how many moles of carbon dioxide can be produced?
Solved
Show answers
More tips
- C Cities and Countries Which Country has the Most Expensive Visa?...
- G Goods and services Don t Let Your Fridge Smell Bother You: How to Get Rid of Unpleasant Odors in Your Refrigerator...
- F Family and Home What does a newborn need?...
- F Food and Cooking How to Make Cottage Cheese Casserole? A Proven Recipe...
- H Health and Medicine How to Improve Eyesight: Science-based Techniques to Enhance Your Visual Acuity...
- A Auto and Moto What to expect from the new VW Golf 7?...
- F Food and Cooking Learn How to Make Ice Cream at Home - Step by Step Guide...
- F Food and Cooking Discover the most delicious spaghetti with these tips...
- F Food and Cooking Why Chicken Liver Pops and How It Can Affect Your Health?...
- F Food and Cooking How to Choose the Right Olive Oil: A Comprehensive Guide...
Answers on questions: Chemistry
- C Chemistry Even though there is an excess of hocl at the end of the reaction, the carbonyl group in camphor is not oxidized to the corresponding acid, why?....
- H History Who is the head of the government in Japan s government? president minister of state prime minister premier...
- M Mathematics Solve for n. round to the nearest tenth, if necessary. 4 7/n=36/54 a. n = 1.3 b. n = 70.5 c. n = 0.7 d. n = 2538...
- M Mathematics HELP ME ASAP I WILL GIVE U HEAD...
- C Chemistry Aphysical property that always stay the same...
- M Mathematics Write an equation for the line that goes through the points (6, -7) and (0, -6). Write the equation in standard form....

Ответ:
1 mole C3H8 5 moles O2 3 moles CO2
4.00 moles C3H8 4.00 moles O2 ( moles CO2)
moles CO2 = 4.00 x 3 / 5
moles CO2 = 12 / 5
= 2.40 moles of CO2
hope this helps!
Ответ:
Answer : The correct option is, 5
Solution : Given,
Mass of nickel-59 = 0.17 g
Mass of cobalt-59 = 5.27 g
Equation for the radioactive decay of nickel-59 is :
Now, we have to calculate the initial amount of nickel-59, we are using the stoichiometry of the reaction and moles of the reactant and product.
Formula used :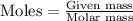
Moles of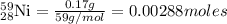
Moles of
By stoichiometry of the reaction,
1 mole of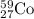 is produced by 1 mole
is produced by 1 mole 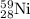
So, 0.089 moles of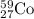 will be produced by =
will be produced by = 
Amount of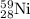 decomposed will be = 0.089 moles
decomposed will be = 0.089 moles
Initial amount of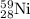 will be = Amount decomposed + Amount left = (0.00288 + 0.089)moles = 0.09188 moles
will be = Amount decomposed + Amount left = (0.00288 + 0.089)moles = 0.09188 moles
Now, to calculate the number of half lives, we use the formula :
where,
a = amount of reactant left after n-half lives = 0.00288 moles
n = number of half lives
Now put all the given values in above equation, we get
Taking log on both sides, we get
Therefore, '5' number of half-lives have passed since the meteorite formed .