alejandraluna95
01.05.2021 •
Chemistry
If you were to go into your kitchen and stir milk into vanilla pudding mix, or cream into coffee, you would be making
a. chemical compound
b. homogeneous mixtures
c. heterogeneous mixtures
d. combinations of elements
Solved
Show answers
More tips
- T Travel and tourism How to Use a Compass: A Beginner s Guide...
- C Computers and Internet Porn Banner: What It Is and How to Get Rid Of It?...
- F Food and Cooking How many stages of coffee roasting are there?...
- F Food and Cooking From Latte to Espresso: Which Coffee Drink is the Most Popular on Earth?...
- F Food and Cooking Experts Name Top 5 Healthiest Teas...
- C Computers and Internet Google Search Tips and Tricks: Everything You Need to Know...
- H Health and Medicine Discover the Hidden Principles and Real Results of the Japanese Diet...
- H Health and Medicine Liver Cleansing - Rejuvenation for the Body?...
- S Style and Beauty Is Photoepilation the Solution to Unwanted Hair Forever?...
- S Style and Beauty Unbelievable Hairstyles for Long Hair - 2021 Trends...
Answers on questions: Chemistry
- C Chemistry What information does an equilibrium constant give about a reaction?...
- C Chemistry What is the formula of the ionic compound formed by magnesium and sulfur (S) atoms....
- C Chemistry A weather balloon is inflated to a volume of 2200 L or 2.2 x 103 L with 374 g of helium. What is the density of helium in g/L...
- C Chemistry How do you think chromatography can be used in finding evolutionary relationships between organisms? HINT: use the colors...
- C Chemistry 2. Why was the balloon important for tracking the movement of matter in your experiment?* S (10 Points) Enter your answer Calendar Files Create Team Activity Hi Teams Assignments...
- C Chemistry How are dogs born? (10 points)...
- C Chemistry In a chemical reaction, mass is conserved. This means that... O A. The mass of the reactants stays the same during a chemical reaction OB. The mass of the products stays the...
- C Chemistry ¿Cortar árboles de un bosque asegurando luego volver a sembrar es una actividad sostenible? es un examen plz...
- C Chemistry 2H2 + O2 → 2H2O PLEASE HELP ASAP Which of the following statements would be correct if one mole of hydrogen was used in this reaction? (5 points) One mole of oxygen was used...
- C Chemistry Silver nitrate reacts with barium chloride. If the reaction is started with 12.6 grams of silver nitrate and 8.4 grams of barium chloride, how many grams of silver chloride...

Ответ:
Explanation: when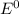 = +ve, reaction is spontaneous
= +ve, reaction is spontaneous
1.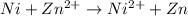
Here Ni undergoes oxidation by loss of electrons, thus act as anode. zinc undergoes reduction by gain of electrons and thus act as cathode.
Where both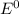 are standard reduction potentials.
are standard reduction potentials.
As the standard emf comes to be negative, the reaction is non spontaneous.
2)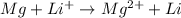
Here Mg undergoes oxidation by loss of electrons, thus act as anode. Lithium undergoes reduction by gain of electrons and thus act as cathode.
As the standard emf comes to be negative, the reaction is non spontaneous.
3)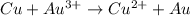
Here Cu undergoes oxidation by loss of electrons, thus act as anode. Gold undergoes reduction by gain of electrons and thus act as cathode.
As the standard emf comes to be positive, the reaction is spontaneous.
4)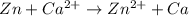
Here Zn undergoes oxidation by loss of electrons, thus act as anode. Calcium undergoes reduction by gain of electrons and thus act as cathode.
As the standard emf comes to be negative, the reaction is non spontaneous.