kenzie19169
30.07.2019 •
Chemistry
In an oxidation–reduction reaction, what happens to the electrons in the oxidation process? electrons are bonded. electrons are gained. electrons are lost. electrons are replaced.
Solved
Show answers
More tips
- G Goods and services How to Choose a Humidifier? Helpful Tips and Recommendations...
- W Work and Career What is the Most In-Demand Profession in the Modern World?...
- A Auto and Moto How Can Parking Sensors Help Drivers?...
- H Health and Medicine What is the Normal Blood Sugar Level in a Healthy Person?...
- F Food and Cooking Red Caviar: How to Choose the Best?...
- C Computers and Internet Clearing Cache: How to Speed Up Your Browser...
- S Style and Beauty How are artificial nails removed?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
- F Food and Cooking Everything You Need to Know About Pasta...
Answers on questions: Chemistry
- C Chemistry Match the term with its description. match term definition colloid a) a mixture in which two or more liquids are mixed that normally are not mixable emulsion b) mixture that...
- C Chemistry The burning of fossil fuels will release into the carbon-oxygen cycle. nitrogen soot oxygen carbon...
- C Chemistry I have 9 protons, 10 neutrons, and 5 electrons. what is the charge of my nucleus?...
- C Chemistry For the reaction 2NO + O2 = 2NO2, what is the maximum amount of NO2 (46.0055 g/mol) which could be formed from 8.6g of NO (30.0061 g/mol) and 1.78g of O2 (31.9988 g/mom)?...
- E English Literacy devices of the poem Certain Choices by Richard Shelton...
- E English Сочинение как я провел лето на английском...
- M Mathematics The circle graph shows the most important problems for the 12,223,017 high school teenagers in a country. Without using a calculator, estimate the number of high school teenagers...
- S Social Studies Define the term mercantile...
- M Mathematics What is the simplified expression for 5 to the power of 4 multiplied by 5 to the power of 3 over 5 to the power of 5...
- M Mathematics Can anyone help me with these i’m not really good at them...

Ответ:
For Plato Users its B. Electrons are gained.
Explanation:
I took the test and got it right
Ответ:
0.044 M is the molarity of 1.60 L of a solution containing 8.53 g of dissolved KBr.
Explanation:
Data given:
volume of the solution = 1.60 litres
mass of KBr = 8.53 grams
atomic mass of KBr = 119.002 gram/moles (K = 39.09 g/mole + 79.9 g/mole)
number of moles =?
molarity of KBr solution =?
First the number of moles will be calculated by using the formula:
number of moles =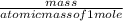
putting the values in the equation:
number of moles =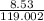
number of moles = 0.071 moles of KBr
now molarity is calculated using the formula:
molarity =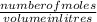
putting the values in the equation:
molarity =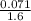
= 0.044 M
molarity of KBr solution is 0.044 M