In aqueous solution the Hg2+ ion forms a complex with four iodide anions. Write the formation constant expression for the equilibrium between the hydrated metal ion and the aqueous complex. Under that, write the balanced chemical equation for the first step in the formation of the complex.
Solved
Show answers
More tips
- F Family and Home How to Teach Your Child to Read?...
- G Goods and services Stock center - a modern way of optimizing logistics...
- F Family and Home Ways to Attract Wealth into Your Home...
- F Food and Cooking Discover How to Properly Prepare Dough for Rasstegai...
- S Style and Beauty Discover the Art of Nail Design: How Do You Paint Your Nails?...
- C Computers and Internet How Much Does an iPhone Cost in America?...
- F Family and Home How to Keep Your Home Warm: Tips and Tricks...
- H Health and Medicine Tick Traps: How to Remove Them Safely and Effectively...
- A Animals and plants 10 Tips for Growing Delicious and High-Quality Tomatoes in Your Garden...
- S Sport How to Choose Tennis Rackets?...
Answers on questions: Chemistry
- C Chemistry Is it better to use graphite or carbon electrodes during electrolysis if I am trying to investigate volume of gas produced ?...
- C Chemistry When iodine is treated with hot aqueous potassium hydroxide a colourless solution is formed.Addition of concentrated sulphuric acid to the solution causes it to turn red brown as iodine...
- C Chemistry C20H16+ _Cl2 → C+ HCl Please help me fast...
- C Chemistry Chemistry class 9 chapter 1...
- C Chemistry How much heat in kilojoules is evolved in converting 2.00 mol of steam at 150 ∘c to ice at -55.0 ∘c? the heat capacity of steam is 1.84 j/g∘c and that of ice is 2.09 j/g∘c. remember...
- C Chemistry Why is reaching activation energy necessary? a)reactants require a minimum amount of energy to start to break a chemical bond. b)products require a minimum amount of energy to start...
- C Chemistry In this lab you observed that each element emits a unique color of light when heated in a flame. if these light emissions were examined through a prism, you would observe that the...
- C Chemistry Im dummy stupid which is an example of unbalanced forces? a vase sitting still on a desk.a person leaning on a wall that is not movinga ball sitting in a field not moving.tug of war...
- C Chemistry Im dummy stupid and i need . true or false. a force is a push or pull exerted on an object.truefalse...
- M Mathematics Will mark brainliest. the dot plots below show the weights of the players of two teams: based on visual inspection of the dot plots, which team appears to have the larger mean weight?...

Ответ:
Following are the solution to the given question:
Explanation:
In the aqueous solution of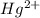 ion is used to represent a
ion is used to represent a ![[Hg(H_2O)_4]^{2+}](/tpl/images/1394/5126/65dfb.png) .
.
It is used to form a complex with iodide ions as![[Hgl_4]^{2-}](/tpl/images/1394/5126/970b6.png) .
.
Conversion reaction:
Following are the formation constants:
Following are the first step to the conversion:
Ответ:
Sifter
Explanation: They are called homogenous mixtures. If you put sand into a glass of water, it is considered to be a mixture. You can always tell a mixture, because each of the substances can be separated from the group in different physical ways. You can always get the sand out of the water by filtering the water away.