gustinya000
02.07.2019 •
Chemistry
In the reaction fecl2 + 2naoh -> fe(oh)2(s) + 2nacl, if 6 moles of fecl2 are added to 6 moles of na0h, how many moles of naoh would be used up in the reaction? - apex a. 3 b. 6 c. 2 d. 1
Solved
Show answers
More tips
- H Health and Medicine 10 Simple and Effective Tips on How to Lose Weight in a Week...
- C Computers and Internet Where did torrents.ru move to?...
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
- G Goods and services How to Choose the Right High Chair for Your Baby?...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
- S Style and Beauty How to braid friendship bracelets?...
Answers on questions: Chemistry
- C Chemistry 20gm of saturated salt solution at 80 degree celcius colled to 30 degree celcius.How much of the salt will crystalized out of its solubilities at 80 degree celsius and 30...
- C Chemistry 5ml of mixture of CH4 and C2H4 need 13.2 ml of oxygen for complete combustion.calculate the volume of each gas in the mixture based on the law of gaseous volume....
- C Chemistry Formulae for covalent compounds sulphur hexaflorideboron trifloridechlorine dioxidehydrogen iodidephosphorous triodidecarbon dioxidecarbon monoxide...
- C Chemistry **help** ................
- C Chemistry - This question is about the electrolysis of silver nitrate solution A. What ions are present in the solution? B. Write the half equations for the each electrode? At the cathode...
- C Chemistry Krypton 81 is used for lung ventilation studies. It’s half life is 13 seconds. How long will it take for 100 cpm to decrease in activity to 25 cpm...
- C Chemistry What is the molarity when 1.25 mol is dissolved in 3.50 L of solution?...
- C Chemistry A voltaic cell is constructed with an Ag/Ag half-cell and a Pb/Pb2 half-cell. The silver electrode is positive. Write the balanced half-reactions and the overall reaction....
- C Chemistry If the [OH1-] of a solution at 25oC is 5.8x10^(-5) mol/L, calculate the [H1+] in mol/L, the pH, and the pOH....
- C Chemistry 2select the correct sequence of steps for lighting a bunsen burner. a. apply the spark above the barrel of the bunsen burner. b. open the gas adjustment at the base of the...

Ответ:
Answer : The correct option is, (B) 6 mole
Explanation :
Given moles of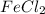 = 6 moles
= 6 moles
Given moles of = 6 moles
= 6 moles
First we have to calculate the limiting and excess reagent.
The balanced chemical reaction is,
From the given balanced reaction, we conclude that
As, 1 moles of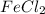 react with 2 moles of
react with 2 moles of 
So, 6 moles of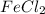 react with
react with  moles of
moles of 
From this we conclude that,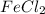 is an excess reagent and
is an excess reagent and  is a limiting reagent because the given moles are less than the required moles and it limits the formation of product.
is a limiting reagent because the given moles are less than the required moles and it limits the formation of product.
Thus, the number of moles of NaOH used up in the reaction = Required moles of NaOH - Given moles of NaOH
The number of moles of NaOH used up in the reaction = 12 - 6 = 6 moles
Therefore, the number of moles of NaOH used up in the reaction will be, 60 moles
Ответ:
Lithium ( 3),Calcium (20),Copper (29),Strontium (38),Potassium (19),Sodium (11)
Explanation: