jeremiah1212
02.07.2019 •
Chemistry
In this experiment, you will titrate a solution of hcl whose concentration is unknown with a solution of naoh whose concentration is known. based on what you just reviewed, express this goal in the form of an investigative question that you can answer by doing the experiment.
Solved
Show answers
More tips
- F Family and Home How to Choose the Perfect Air Conditioner for Your Life...
- H Health and Medicine Discover the Hidden Principles and Real Results of the Japanese Diet...
- H Health and Medicine Understanding Pregnancy Tests: What You Need to Know?...
- H Health and Medicine What Makes a Man a Man?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
Answers on questions: Chemistry
- C Chemistry We are slowly learning that even if we react, it won t change anything, it won t make people suddenly love and respespect us, it won t magically change their minds. Sometimes...
- C Chemistry What part of the atom contains the protons and neutrons?...
- C Chemistry Lord kokichi plz help what properties influence weather...
- C Chemistry What are the correct products for the following reaction? K2SO4 + MgF2 Question 3 options: 2KF + MgSO4 F2SO4 + K2Mg No reaction will occur K2SO4 + F2Mg...
- C Chemistry I need help with this question! Can someone answer it for me? thank you!...
- C Chemistry Calculate the number of moles of HCl in 62.85 mL of 0.453 M solution of HCl....
- C Chemistry What law allows calories to be determined by heat (energy) transfer from one substance to another, but it is never destroyed?...
- C Chemistry A 0.5 M solution contains 20 grams of solute in 500 milliliters of solution. What is the mass of 1 mole of the solute? A. 10 g B. 20 g C. 40 9 D. 80 g...
- C Chemistry Which would increase the amount of dissolved oxygen in a pond? a. removing plants. b. removing fish. c. adding plants. d. both b and c are correct....
- C Chemistry Which of the following would most likely occur if the oxygen in a pond decreased? a. fish would grow larger. b. fish would die. c. the temperature of the pond would decrease....

Ответ:
Here we have to get the equation or process by which the unknown concentration of HCl can be determined by the known concentration of NaOH.
The equation to determine the unknown concentration will be ×
× 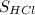 =
=  ×
×  .
.
The volume of the HCl or titrant will be known as it will be taken in the conical flask and the volume of the titre i.e. NaOH will be taken in burret. The indicator used in this titration is phenlopthalien.
At the end point the color of the colorless solution will become light pink and from the burret reading we can get the volume of NaOH required in the titration process.
On plugging the volume, strength of NaOH and volume of HCl, we can obtain the strength of HCl.
Ответ:
B--StartFraction pi Over 2 EndFraction radians
Step-by-step explanation: