uticabadgirl
16.11.2019 •
Chemistry
In water solutions of hcl the free h+ ion is
a. present in very large quantities
b. present in moderate quantities
c. present in small quantities
d. not present
Solved
Show answers
More tips
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- S Style and Beauty How to Get Rid of Peeling Nails: Natural Remedies...
- S Science and Technology Understanding Magnetic Storms: A Guide to This Natural Phenomenon...
- F Family and Home What is Most Important in Men s Lives?...
- G Goods and services Which TV is better - LCD or Plasma?...
- C Computers and Internet Are there special gaming mice?...
- G Goods and services LED-подсветка в LCD-телевизорах: 5 причин, почему она лучше других технологий...
- C Computers and Internet Keep Your Mouse Pad Clean: The Right Way to Clean It?...
Answers on questions: Chemistry
- M Mathematics Good night all ✨ must accompany step by step ! :)...
- M Mathematics Mike needs to paint several rooms in his house. he knows that the larger the rooms are, the more paint he will need....
- E English The welcoming smile of my librarian the anticipation in my heart all those books—another world—just waiting at my fingertips the implicit details in this excerpt show that the speaker...
- M Mathematics Solve each equation for the variable 7x-2=40...

Ответ:
See figure 1
Explanation:
For this question, we have to remember that in the lewis structures all atoms must have 8 electrons. And each atom would have a different value of valence electrons:
Carbon => 4
Oxygen=> 6
Hydrogen=> 1
Additionally, for the hybridizations we have to remember that:
With this in mind, the formaldehyde and formic acid would have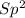 carbons and the ethanol an
carbons and the ethanol an  carbon.
carbon.
Finally, for the oxidation state. We have to remember that if we have more bonds with oxygen, we will have more oxidation. Therefore, the carbon that has more oxidation is the one in the formic acid (we have several bonds with oxygen).
See figure 1
I hope it helps!