In which type of chemical reaction are electrons transferred?
1) organic addition
2) oxidation-reduction
3) double replacement
4) acid-base neutralization
Solved
Show answers
More tips
- A Animals and plants How to Properly Care for a Pet Decorative Rabbit at Home?...
- G Goods and services What Useful Foods Can You Buy at Supermarkets?...
- F Food and Cooking How to Determine Healthy, Nutritious Food for Yourself?...
- A Art and Culture The History and Characteristics of Jazz Bands: A Deep Dive...
- W Work and Career Can Skill Alone Make You a Professional?...
- F Family and Home Daughter says: If you don t want to do it, don t do it. Should we persuade her?...
- S Science and Technology How to Secure Exam Sessions: Silence Mobile Phones in the Classroom...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- P Philosophy Is Everything We Strive for Eventually Achieved and Destroyed?...
- S Society and Politics Understanding Politics and Its Role in the Development of Civilization...
Answers on questions: Chemistry
- M Mathematics I NEED HELP ON NUMBER 6 PLEASE HELP!!...
- S Social Studies German surrender summary...
- M Mathematics Sarah took the advertising department from her company on a round trip to meet with a potential client. Including Sarah a total of people 13 took the trip. She was...
- H History 6 Based on Lincoln s words in the Gettysburg Address, what might he have thought of the peace monument if he were alive to see it? Support your answer with evidence...

Ответ:
In oxidation-reduction reactions (or redox reactions), a number of electrons are transferred between two species. During this process, the oxidation number of an atom (or molecule or ion) changes by receiving or losing an electron.
Ответ:
Step-by-step explanation:
First of all let us have a look at 3 formulas:
Both the formula can be applied to the expression(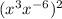 ) during the first step while solving it.
) during the first step while solving it.
Applying formula (1):
Comparing the terms of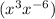 with
with 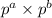
So,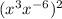 is reduced to
is reduced to 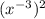
Applying formula (2):
Comparing the terms of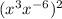 with
with 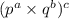
So,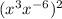 is reduced to
is reduced to 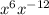 .
.
So, the answers can be: