genyjoannerubiera
14.09.2019 •
Chemistry
Indicate whether each of the following statements is true or false. (3 pts) always use the exact calculated volume of solvent when purifying a solid by recrystallization. when recrystallizing a sample, you should always add as much solvent as needed to dissolve all of the solid present just before filtering the product, the slurry containing the crystals should be cooled in an ice bath that contains only crushed ice.
Solved
Show answers
More tips
- C Computers and Internet How to Properly Repartition a Hard Drive?...
- A Auto and Moto What Is the Cost of Customs Clearance for a Car in Russia?...
- L Leisure and Entertainment Should You Buy a Ceramic Knife?...
- C Computers and Internet How to easily and quickly disable Firebug in Gmail and Google Docs...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Chemistry
- C Chemistry What are the two flasks for measuring the liquid volume?...
- M Mathematics Q(a+1) - 2q(a) if q(x) = x²+3x+4...
- C Chemistry QUESTION 2 1. In a covalent bond between Cl and H, tends to attract electron density from the other element in the bond. Enter the chemical symbol for your answer (case sensitive)....
- M Mathematics Simplify: 17b + 82c + 90 + e - 18 - 10c - b...
- M Mathematics It takes 4 3/8 cups of cheese,7/8 cups of olives,and 2 5/8 cups of sausage to make a signature pizza. how much cheese is needed to make 10 pizzas?...

Ответ:
In this given case, he volume of hydrogen gas produced = 0.03969 L
Given:
0.52 grams - solid magnesium
Solution:
moles of Mg
=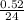
= 0.216 x 10⁻³
Reaction -
Mg + 2HCl -----------------> MgCl2 + H2
1mol 1mol
0.216 x 10⁻³ 0.216 x 10⁻³ mol
moles of H2
= 0.216 x 10⁻³
T = 273 K
P =1 atm
From idea gas P V = n R T
Placing given and known values in ideal gas
1 x V = 0.216 x 10⁻³ x 0.0821 x 273
V = 0.048 L
Thus, the volume of hydrogen gas produced = 0.03969 L
Learn more:
link