Knowing that the molar mass of the compound is 46.069 grams/mole, use the formula weight calculated in part C to
determine the molecular formula.
Select the correct answer from each drop-down menu.
С_H_O_
Solved
Show answers
More tips
- S Science and Technology Exploring Our Galaxy: How Many Planets are in the Milky Way?...
- S Science and Technology Colliders: How They Work and Why They Matter...
- A Animals and plants Unraveling the Mystery of Loch Ness: What Does the Loch Ness Monster Look Like?...
- L Leisure and Entertainment How Many Seasons are There in the TV Show Interns?...
- S Sport Playing Bowling: Rules and Advice for Novices...
- L Leisure and Entertainment The Best Film of 2010: A Look Back at the Academy Awards...
- S Sport How to Learn Swimming? Simple Tips for Beginners...
- C Computers and Internet What is Web 2.0 and How Does it Work?...
- C Computers and Internet War of Social Media: Which Platform is the Leader?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
Answers on questions: Chemistry
- S Social Studies Amineral that is commonly refined in south asia and then exported is a. oil b. ore c. coal d. lumber...
- M Mathematics PLZ HELP?!?!! Tom s boat can travel a mile in 3.2 minutes. How long will it take him to go 67.5 miles? (Give your answer in hours)...
- C Chemistry A schematic diagram of the sublimation process...
- F French Which verb correctly completes this conversation? Lucas: Bonjour. J’ai un rendez-vous avec le médecin. Réceptionniste: Bonjour. , s’il vous-plaît. Le médecin arrive dans 2...

Ответ:
C2H6O.
Explanation:
Ответ:
Explanation:
The given data is as follows.
Density of vinegar = 1.0 g/ml
Specific heat capacity = 4.25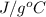
Relation between enthalpy and specific heat is as follows.
Hence, putting the values into the above formula as follows.
= (as density =
(as density = 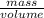 )
)
= - 315 J
Thus, we can conclude that the enthalpy of reaction is -315 J.
As the value is negative so, it means that heat is releasing. Hence, the reaction is exothermic in nature.