Markus was doing his chemistry homework and needed to find the
number of electrons in an atom of carbon. He looked at his periodic table
and saw that it had an atomic mass of 12 and the atomic number was 6.
What should his answer be?
Solved
Show answers
More tips
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
- S Sport When and Where Will the 2014 World Cup be Held?...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- C Computers and Internet How to Learn to Type Fast?...
Answers on questions: Chemistry
- M Mathematics 3. Armani has $7.00 in her purse consisting of nickels, dimes, and quarters. The total number of coins she has is 50. She has twice as many dimes as quarters. Write the...
- E English Preston is writing an essay on consumerism. in this draft, how does he explain the effects of consumerism?...
- A Arts Say something nice about me...
- H History Why is king charles of martel important in europe’s history?...

Ответ:
1869, the Russian chemist Dmitri Mendeleev came to prominence with his tabular ... He found that few of the new developments in the field of chemistry had made ... He corrected the known atomic masses of some elements and he used the ... Atomic number 101, Mendelevium, is named after the periodic table's architect
Explanation:
1869, the Russian chemist Dmitri Mendeleev came to prominence with his tabular ... He found that few of the new developments in the field of chemistry had made ... He corrected the known atomic masses of some elements and he used the ... Atomic number 101, Mendelevium, is named after the periodic table's architect
Ответ:
Explanation:
Moles of = 0.35 mole
= 0.35 mole
Moles of = 0.40 mole
= 0.40 mole
Volume of solution = 1.00 L
Initial concentration of =
= 
Initial concentration of =
= 
Equilibrium concentration of =
= 
The given balanced equilibrium reaction is,
Initial conc. 0.35 M 0.40 M 0 M 0M
At eqm. conc. (0.35-x) M (0.40-x) M (x) M (x) M
Given: (0.35-x) = 0.19
x= 0.16 M
The expression for equilibrium constant for this reaction will be,
Now put all the given values in this expression, we get :
Thus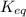 at the temperature of the experiment is 0.56.
at the temperature of the experiment is 0.56.