pamelperezz26
03.12.2020 •
Chemistry
MY ACCOUNT WILL BE DELETED BEFORE MORNING SO HERE ARE MY POINTS I WORKED REALLY HARD FOR THEM SO DONT WASTE THEM ALL IN ONE PLACE:)))
Solved
Show answers
More tips
- H Health and Medicine What are Electronic Cigarettes? The Ultimate Guide...
- C Computers and Internet How to Get Rid of 3pic Infector: Everything You Need to Know...
- A Auto and Moto Experience the World of the Most Expensive Cars on the Planet...
- S Style and Beauty How to Get Rid of a Double Chin?...
- F Food and Cooking How to Cook Julienne? Recipes and Tips...
- D Dating, Love, Relationships 10 Useful Tips on How to Survive a Breakup?...
- F Food and Cooking Apple Cider Vinegar: The Ultimate Health and Beauty Solution...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
Answers on questions: Chemistry
- P Physics 19. A stone has a mass of 390 g and a density of 2.7 g/cm3. Cooking oil has a density of 0.90 g/cm². Which mass of oil has the same volume as the stone? A 130 g B 160...
- M Mathematics Trinity! math skills needed here lol...
- M Mathematics The distance between two stations is 240km. A train takes 4hrs to cover the distance. Calculate the speed of the train...
- M Mathematics The town of Hillsville has a triangular walking trail, as shown on the left. Perry wants to construct a similar trail in his town, but he has only enough space for a...
- M Mathematics Rhonda had $300 to buy supplies for the animal shelter. She bought 8 bags of dog food and 11 leashes. Each bag of food cost $25.19, and each leash cost $4.65. How much...

Ответ:
Thx
Explanation:
Ответ:
Omg thank you so much
Explanation:
Ответ:
No precipitate is formed by reaction of and HCl.
and HCl.
Further Explanation:
Precipitation reactions:
Such reactions involve formation of insoluble salt when solutions of soluble salts combine with each other. This insoluble salt is termed as precipitate. Due to formation of precipitates, these reactions are known as precipitation reactions. These reactions are essential tool in determination of different ions existing in particular solutions.
Reaction between and HCl occurs as follows:
and HCl occurs as follows:
In this reaction, one mole of reacts with one mole of HCl and one mole of
reacts with one mole of HCl and one mole of 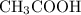 and one mole of NaCl are produced. Both products are soluble in water. Since both of the products of this reaction are water soluble, no precipitate can be formed by reaction of
and one mole of NaCl are produced. Both products are soluble in water. Since both of the products of this reaction are water soluble, no precipitate can be formed by reaction of  and HCl.
and HCl.
Learn more:
1. Balanced chemical equation:
2. Oxidation and reduction reaction:
Answer details:
Grade: Senior School
Subject: Chemistry
Chapter: Chemical reactions and equations
Keywords: NaCH3COO, HCl, CH3COOH, NaCl, precipitate, soluble salts, insoluble salts, no precipitate, salt, weak acid, strong acid, sodium, sodium acetate salt, reaction, acetic acid, sodium chloride, ions, solutions, water soluble.