endermss6220
30.10.2019 •
Chemistry
Need this now !
two more time to go the distance
take less time to go the same distance. the one that is moving will go faster?
a: take more time to go the distance
b: take less time to go the same distance
c: take the same time as the slower object
d: none of the above
Solved
Show answers
More tips
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- L Leisure and Entertainment What to Bring on a Hike? Essential Items to Pack for a Safe and Enjoyable Adventure...
- L Leisure and Entertainment Couchsurfing: A New Way to Travel...
- S Style and Beauty Autotanning: Harmful or Safe?...
- F Food and Cooking 10 Ideas for a Wedding Anniversary Gift...
- H Health and Medicine How to Reduce Sweating in the Heat and Beyond: Say Goodbye to Excessive Sweat...
- F Food and Cooking Do Aphrodisiacs Really Work? Separating Fact from Fiction...
- H Health and Medicine What to Eat to Lose Weight?...
- A Animals and plants How to Teach Your Parrot to Talk?...
- H Health and Medicine How to Improve Your Posture?...
Answers on questions: Chemistry
- C Chemistry Write the reaction between Hydrochloric acid and Sodium Hydroxide, and Explain what the endpoint of the reaction is for the reaction and what is important about that...
- C Chemistry PLEASE HELP Which of the following correctly describes the process of water freezing? A The ordered liquid water molecules become more ordered as they become ice....
- C Chemistry The temperature of 568.9 mL of N20 is changed from -34.64 Celsius to 12.86°C during this the pressure also changes from .3002 atm to 4.0884 atm what is the new volume...
- C Chemistry What is the half-life of a radioisotope if a 40 g sample becomes 10 g after 20 minutes?...
- C Chemistry What does the hydrosphere include? Glaciers Groundwater Lava Polar Ice...
- C Chemistry How many formula units are in 0.96 moles of H2O...
- C Chemistry Normal text Times New... 14 + BIO GD 1 2 3 4 5 6 3. Acetylene gas, CH,, is used in welding, produces an extremely hot flame when it burns in pure oxygen according...
- C Chemistry You are given a solid that is a mixture of na2so4 and k2so4. a.205g sample of the mixture is in water. an excess of an aqueous solution of bacl2 is added. the baso4...
- C Chemistry An ideal gaseous reaction (which is a hypothetical gaseous reaction that conforms to the laws governing gas behavior) occurs at a constant pressure of 50.0 atm and...
- C Chemistry Which reactions are redox reactions? check all that apply. check all that apply. 4li(s)+o2(g)→2li2o(s) mg(s)+fe2+(aq)→mg2+(aq)+fe(s) pb(no3)2(aq)+na2s?...

Ответ:
There are 1.393 x 10²⁴ atoms in 25.00 g of B.
Explanation:
Hey there!
We are given a value, in grams, that we need to convert to a number of atoms.
We can convert grams to atoms by using Avogadro's Number ( ). This number is equivalent to
). This number is equivalent to 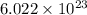 .
.
This number can be used to convert any values to:
atomsmoleculesformula unitsmolesIn order to do this problem, we will need to use dimensional analysis (DA). This process allows us to convert from grams to atoms.
We need to set up our ratios in order to work this out. We can use a periodic table to help us through this next part of the problem.
1. Locating the number of moles of B in the sample
We first need to find the amount of moles of boron (B) there are in the sample.
Checking a periodic table, the atomic mass in atomic mass units (amu) is 10.81 amu.
Atomic mass units can easily be converted to grams and these units can be used interchangeably.Therefore, for each atom of boron, it weighs 10.81 grams to us. This is equivalent to the mass of one mole of boron.
To find the number of moles, we have two possible ratios we can use:
These ratios mean the same thing, but we need to convert our final unit to moles.
We are given a sample in grams, and when dividing our units, we need to keep moles.
Since the first portion of our expression is in grams, we need to have grams in the bottom of our expression.
We can now simplify the expression. Our grams B unit will cancel out, so we are therefore left with moles B remaining.
2. Locating the number of atoms in the sample
Now with our equation, we can convert our number of moles that would be solved if we stopped with the above. However, we need to convert to atoms.
We use Avogadro's number and create a ratio with that of moles.
We need to cancel out our moles and end with atoms, so we must have moles in the denominator. Therefore, we use the first ratio.
Using our previous expression, we multiply by this new ratio and solve the expression.
This expression can now be operated. You will need a calculator to perform this calculation.
Our numerator is:
Plugging this into a calculator, we get:
Our denominator is:
This simplifies to:
Dividing our numerator and denominator:
Plugging this into a calculator, we get:
3. Simplifying with significant figures
Now, we need to take into account that we have significant figures. We are given this original value:
This value has four significant figures, which means we need to round our value we received above to four significant figures.
Our units are added as well as our scientific notation:
Therefore, our final answer is choice A.