ggdvj9gggsc
02.08.2019 •
Chemistry
Nitric acid and zinc react to form zinc nitrate, ammonium nitrate, and water. how many atoms of zinc react with 1.49g hno3? calculate the number of grams of zinc that must react with an excess of hno3 to form 29.1g nh4 no3.
Solved
Show answers
More tips
- L Leisure and Entertainment 10 Ways to Celebrate Your Favorite Holidays: A Guide to Festivities and Traditions...
- S Sport When will the Biathlon World Championships 2011 take place in Khanty-Mansiysk? Answers to frequently asked questions...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
Answers on questions: Chemistry
- S Social Studies Any body interested in zoom meetingpurpose: friendship...
- M Mathematics 5) Find the numbers: a. 0.75 of what number is 9? b. 0.8 of what number is 8?...
- C Chemistry How much energy is given off by the following reaction, if 162.5 g of oxygen reacts with 216.7 g of ammonia (NH3)? (16 pts) 4 NH3 + 5 O2 4 NO + 6 H2O ΔH = -1225.6 kJ...
- W World Languages Explain why you agree or disagree that emotional health can affect our physical health...

Ответ:
4 mol of Zn react with 10 mol of HNO3. So:
4 x 6.0x10^23 atoms of Zn 630 g of HNO3
x 1.35 g
x= 5.1x10^21 atoms of Zn.
b)
Molar mass of Zn: 65.37 g/mol
Molar mass of NH4NO3: 80 g/mol
4 mol of Zn form 1 mol of NH4NO3. So:
4 x 65.37g of Zn 80 g of NH4NO3
m 27.5 g
m= 89.9 g of Zn.
Ответ:
For 1: The number of atoms of zinc are atoms.
atoms.
For 2: Mass of zinc reacted are 95.1279 grams.
Explanation:
For the given chemical reaction, the equation follows:
To calculate the number of moles, we use the equation:
 ....(1)
....(1)
For 1:For nitric acid:
Mass of nitric acid = 1.49 grams.
Molar mass of nitric acid = 63 g/mol
Putting values in equation 1, we get:
By Stoichiometry of the reaction:
10 moles of nitric acid reacts with 4 moles of zinc metal.
So 0.0236 moles of nitric acid will react with =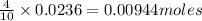 of zinc metal
of zinc metal
Now, according to Mole concept:
1 mole of an element contains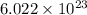 number of atoms.
number of atoms.
So, 0.00944 moles of zinc will contain number of atoms.
number of atoms.
Hence, the number of atoms of zinc are atoms.
atoms.
For 2:For ammonium nitrate:
Given mass of ammonium nitrate = 29.1 grams
Molar mass of ammonium nitrate = 80 g/mol
Putting values in equation 1, we get:
By Stoichiometry of the given reaction:
1 mole of ammonium nitrate is produced from 4 moles of zinc.
So, 0.36375 moles of ammonium nitrate will be produced from = of zinc.
of zinc.
Now, calculating the mass of zinc.
Moles of zinc = 1.455 moles
Molar mass of zinc = 65.38g/mol
Putting values in equation 1, we get:
Hence, mass of zinc reacted are 95.1279 grams.
Ответ:
It would be the innate immune system or just the immune system
Explanation:
:)