arianna23717
01.07.2019 •
Chemistry
Nitrogen dioxide is a red-brown gas responsible for the brown color of smog. a container of nitrogen dioxide that is at low pressure and at room temperature has a volume of 3.41l. after more nitrogen dioxide is added, the container holds 2.28×10−2mol of nitrogen dioxide and the volume of the container is 5.11l, still at the same pressure and temperature. how many moles of nitrogen dioxide were in the container initially? give your answer in three significant figures.
Solved
Show answers
More tips
- F Food and Cooking Ginger: A Universal Ingredient for Various Dishes...
- A Auto and Moto Which alarm system to choose?...
- C Computers and Internet Porn Banner: What It Is and How to Get Rid Of It?...
- F Food and Cooking How many stages of coffee roasting are there?...
- F Food and Cooking From Latte to Espresso: Which Coffee Drink is the Most Popular on Earth?...
- F Food and Cooking Experts Name Top 5 Healthiest Teas...
- C Computers and Internet Google Search Tips and Tricks: Everything You Need to Know...
- H Health and Medicine Discover the Hidden Principles and Real Results of the Japanese Diet...
- H Health and Medicine Liver Cleansing - Rejuvenation for the Body?...
- S Style and Beauty Is Photoepilation the Solution to Unwanted Hair Forever?...
Answers on questions: Chemistry
- C Chemistry Meteors look like they have tails. Are they the same as comet tails?...
- C Chemistry 100 POINTS- Which organelle could best be compared to a closet in a house? A. chloroplast B. mitochondrion C. nucleus D. vacuole...
- C Chemistry If you touch an electric wire and get a shock, what can you infer about the resistance of your body compared to the resistance of the circuit?...
- C Chemistry A solution consisting of 0.210 mol of methylbenzene, C6H5CH3, in 248 g of nitrobenzene, C6H5NO2, freezes at 0.1°C. Pure nitrobenzene freezes at 6.0°C. What is the...
- C Chemistry A solution is prepared by dissolving 0.23 mol of lactic acid and 0.27 mol of sodium lactate in water sufficient to yield 1.00 L of solution. The addition of 0.05...
- C Chemistry for the reaction represented by the equation C+ 2H2 - CH4 how many moles of hydrogen are required to produce 10 mol of methane, CH4...
- B Biology Answer asap! if an astronaut walked on mars without wearing her spacesuit, would she die first from suffocation, boiling fluids, freezing, or something else?...
- M Mathematics After recording the pizza delivery times from 2 different shops you concluded that one pizza shop has a mean of 46 minutes with a standard deviation of 3 minutes,...
- E English Read the passage from “the monkey’s paw.” the whites are being offered compensation for herbert’s death. in what way does the stranger’s answer increase the tension...
- M Mathematics How do u write 4 less than b is equal to 44...

Ответ:
Initially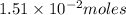 of nitrogen dioxide were in the container .
of nitrogen dioxide were in the container .
Explanation:
Volume of the container at low pressure and at room temperature =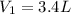
Number of moles in the container =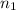
After more addition of nitrogen gas at the same pressure and temperature.
Volume of the container after addition =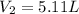
Number of moles in the container after addition=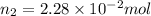
Applying Avogadro's law:
Initially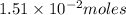 of nitrogen dioxide were in the container .
of nitrogen dioxide were in the container .
Ответ:
B
Explanation: