On a balance, you have beakers of AgNO3AgNO3 solution and NaClNaCl solution. When mixed, they will form AgCl(s)AgCl(s). What will happen to the mass of the new mixture?
Solved
Show answers
More tips
- G Goods and services Is an Air Ionizer Necessary for Your Home?...
- C Construction and repair How to Choose the Best Underfloor Heating?...
- C Computers and Internet How to Get Rid of Windows Genuine Check?...
- C Computers and Internet War of Social Media: Which Platform is the Leader?...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
Answers on questions: Chemistry
- C Chemistry Complete the following question: the more dense the liquid, the higher the at a particular depth....
- C Chemistry The molecular mass for O2(g) is 32.0. By experiment a student measured the mass of O2(g) to be 28.0. What is the percent error in the student s result? A) 1.25% C) 12.5% B)...
- C Chemistry Can anyone break down itjust like 2nd picture and send me plz...
- C Chemistry Can someone please help me it s urgent! Write a statement about each pair of lines in this construction. (3 statements)...
- C Chemistry If the substance in the diagram was heated to t2, what is happening if the substance is allowed to approach t1? select all that apply. the average kinetic energy is decreasing....
- C Chemistry Alightweight material is being tested for effectiveness in blocking ultraviolet (uv) rays from the sun that cause skin to burn and possibly develop skin cancer. which method...
- C Chemistry One characteristic of a stable ecosystem is...
- C Chemistry Unknown element x is a metal that ionically bonds to sulfur.is the formula, x3s feasible? why or why not? a)it is feasible. the three metallic ions each receive an electron...
- C Chemistry By how much will the ph change if 0.025 mol of hcl is added to 1.00 l of the buffer that contains 0.15 m hc2h3o2 and 0.25 m c2h3o2–? use ka = 1.8 × 10–5 for hc2h3o2....
- C Chemistry Calculate the mole fraction of total ions in an aqueous solution prepared by dissolving 0.400 moles of mgcl2 in 850.0 g of water....

Ответ:
Explanation:
According to the law of conservation of mass, mass can neither be created nor it can be destroyed. But it can be simply transformed from one form to another.
Therefore, when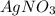 is added to NaCl then the compound formed will have same mass as that of reactants.
is added to NaCl then the compound formed will have same mass as that of reactants.
Total mass of reactants is (169.87 + 58.44) g/mol = 228.31 g/mol
Total mass of products is (143.32 + 84.99) g/mol = 228.31 g/mol
Thus, we can conclude that mass of the new mixture will stay the same.
Ответ:
amine
Explanation:
Carboxylic acid is an acid, so it would require a base like amine to neutralize it.