kutemigos9211
12.05.2021 •
Chemistry
On a hot day, a 15.0 kg window increased from 20.0 degrees C to 26.7 degrees C. How much heat energy did the glass window absorb? (specific heat of glass = 0.840 J/gC)
Solved
Show answers
More tips
- A Animals and plants 10 Tips for Growing Delicious and High-Quality Tomatoes in Your Garden...
- S Science and Technology Colliders: How They Work and Why They Matter...
- S Science and Technology Exploring Our Galaxy: How Many Planets are in the Milky Way?...
- W Work and Career Everything You Need to Know About MBA Programs...
- B Business and Finance What is the Difference Between Visa and Visa Gold?...
- C Computers and Internet The Twitter Phenomenon: What it is and How to Use it...
- S Sport When and Where Will the 2014 World Cup be Held?...
- S Sport When is the Champions League final?...
- C Construction and repair How to Choose the Best Underfloor Heating?...
- A Animals and plants How ants survive winter: exploring the secrets of their winter life...
Answers on questions: Chemistry
- M Mathematics a map has a scale of 2 in.=25 mi.town A 135 miles from town B.what equation can be used to calculate the distance between the two towns on the map...
- F French VI Complete the following sentences by a) placing the adjectives provided in the appropriate place. b) make the necessary agreements (9 pts) Modèle: Marie est une étudiante( travailleur...
- H History What is a historian in the michigan constitution so what is a historian...
- E English What words are clues that this story involves aspects of french culture? a) the words used for money are indicative of the french culture. b) the phrase three-day old cloth is...
- C Chemistry How many moles of chlorine are in 100g Chlorine (CI^2)? A) 64.6B) 100C) 1.41 D) 0.355...

Ответ:
The heat energy absorbed by glass window is 84420 J.
Explanation:
Given: Mass = 15 kg (1 kg = 1000 g) = 15000 g
Initial temperature =
Final temperature =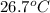
Specific heat capacity =
Formula used is as follows.
where,
q = heat energy
m = mass of substance
C = specific heat capacity
Substitute the values into above formula as follows.
Thus, we can conclude that the heat energy absorbed by glass window is 84420 J.
Ответ:
the tendency toward a relatively stable equilibrium between interdependent elements, especially as maintained by physiological processes.
Explanation: