xoxotrish5401
19.02.2020 •
Chemistry
One mole of an ideal gas doubles its volume in a reversible isothermal expansion. (a) What is the change in entropy of the gas? (b) If 1500 J of heat are added in this process, what is the temperature of the gas?
Solved
Show answers
More tips
- S Sport How to Choose Tennis Rackets?...
- H Health and Medicine AKDS Vaccination: Ensure Your Child s Safety...
- H Health and Medicine Naskol ko Opasen Ukus Kleshcha i Kak Ego Raspoznat...
- C Computers and Internet How to Delete Your Account on Odnoklassniki...
- H Health and Medicine What to Do When Your Jaw Locks Up?...
- G Goods and services What Are the Most Popular Services?...
- P Philosophy How did the concept of module arise in computer science?...
- F Food and Cooking How to Cook Julienne? Recipes and Tips...
Answers on questions: Chemistry
- E English What poetic device is burble...
- M Mathematics Two fair dice are tossed. a. what is the probability that the sum of the number of dots shown on the upper faces is equal to 7? to 11? b.what is the probability that you...
- H History What was the working conditions in many of the factories in europe and the u.s. in the 2nd industrial revolution? describe the living conditions in the 2nd industrial revolution....
- M Mathematics Based on the table which best predicts the end behavior of the graph f(x)?...

Ответ:
Explanation:
The given data is as follows.
n = 1 mol,
Q = 1500 J, R = 8.314 J/mol k
(a)
And, according to the first law of thermodynamics
And, in an isothermal process the change in internal energy of the gas is zero.
Hence, 0 = Q - W
or, W = Q
Expression for work done in an isothermal process is as follows.
W =
As W = Q, Hence expression for Q will also be given as follows.
Q =
Now,
[/tex]\Delta S = nR ln \frac{V_{f}}{V_{i}}[/tex]
=
= nR ln 2
=
= 5.76 J/K
Therefore, change in entropy is 5.76 J/K.
(b) As, Q =
=
= nRT ln 2
T =
=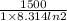
= 260.4 K
Therefore, temperature of the gas is 260.4 K.
Ответ: