drealtania21
07.04.2021 •
Chemistry
pada pendapat anda,apakah cara yang berkesan untuk menerapkan ada,budaya,nilai nilai murni dalam kalangan generasi masa ini?
Solved
Show answers
More tips
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
- S Sport When and Where Will the 2014 World Cup be Held?...
- C Computers and Internet How to Choose a Monitor?...
Answers on questions: Chemistry
- C Chemistry Why is it rare for large meteors to crash into the Earth s surface?...
- M Mathematics Angl is a square. The equation of line NG is Y= 1/2 X - 6. Find the equation of line LG in slope intercept form given that the coordinates of L are (-5,1)...
- M Mathematics In the equation below, what is the coefficient of the variable? 26=9x...
- C Computers and Technology What is the shortcut for the following a hyperlink in word...
- B Biology Without enzymes, the chemical reactions in your body would a. happen too fast b. occur too slowly to benefit the cell c. require a different ph d. occur at much the same rate as they...
- E English Why does Jonas want Gabriel in his room? The Giver pages: 108-117 Chapter 14 PLS HELP ONLY IF YOU HAVE READ THE BOOK...

Ответ:
wht
Explanation:
Ответ:
Dapat mahalin ng mundo ang bawat isa at magkalat ng kapayapaan sa halip na gumamit ng poot
Ответ:
The empirical formula : C₁₂H₁₇N₂O₄P
Further explanationThe empirical formula is the smallest comparison of atoms of compound =mole ratio of the components
The principle of determining empirical formula
Determine the mass ratio of the constituent elements of the compound. Determine the mole ratio by by dividing the percentage by the atomic mass C
H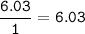
N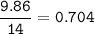
O
PDivide by the smallest mole ratio(0.352)
C : H : N : O : P