Tianylee2328
16.07.2021 •
Chemistry
Please explain how to do it as well!
Write a complete, balanced equation for the following reactions:
a) The combustion of C₆H₁₂O (teachers note: You figure out products).
b) Aqueous ferric iron (III) sulfate plus barium hydroxide (teachers note: You figure out the products).
Solved
Show answers
More tips
- F Food and Cooking Ginger: A Universal Ingredient for Various Dishes...
- A Auto and Moto Which alarm system to choose?...
- C Computers and Internet Porn Banner: What It Is and How to Get Rid Of It?...
- F Food and Cooking How many stages of coffee roasting are there?...
- F Food and Cooking From Latte to Espresso: Which Coffee Drink is the Most Popular on Earth?...
- F Food and Cooking Experts Name Top 5 Healthiest Teas...
- C Computers and Internet Google Search Tips and Tricks: Everything You Need to Know...
- H Health and Medicine Discover the Hidden Principles and Real Results of the Japanese Diet...
- H Health and Medicine Liver Cleansing - Rejuvenation for the Body?...
- S Style and Beauty Is Photoepilation the Solution to Unwanted Hair Forever?...
Answers on questions: Chemistry
- E English Please help me with this homework...
- B Biology Some mercury-resistant bacteria can convert toxic forms of mercury to less toxic forms using the mer operon under transcriptional regulation the merr protein. what...
- H History How do political parties in the us help citizens engage with their government...
- E English How many days does the Time Traveller report that he’s been travelling in The Time Machine? A. 4 B. 3 C. 8 D. 7...

Ответ:
a.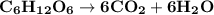
△H=−72 kcal
The energy required for production of 1.6 g of glucose is [molecular mass of glucose is 180 gm]
b.
The iron(III) ions and chloride ions remain aqueous and are spectator ions in a reaction that produces solid barium sulfate.
Ответ:
in front of the elements
Explanation:
The coefficient tells you how much of each element there is in a molecule.
Hope that helps.