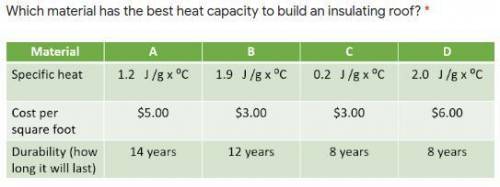
Please help!! chemistry
Solved
Show answers
More tips
- F Food and Cooking What Foods Can Nursing Moms Eat?...
- L Leisure and Entertainment Choosing the Right Books to Read: Tips and Recommendations...
- L Leisure and Entertainment Couchsurfing: A New Way to Travel...
- G Goods and services Which TV is better - LCD or Plasma?...
- S Sport How to Learn to Pull Up on Monkey Bars?...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- C Computers and Internet Where did torrents.ru move to?...
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
Answers on questions: Chemistry
- C Chemistry Determine the vapor pressure (atm) of rubbing alcohol (isopropanol) at 20.0 °c. the normal boiling point of isopropanol is 82.3 °c and the heat of vaporization (δhvap) is...
- C Chemistry The streams and smaller rivers that feed into the main river are its a- watersheds b- tributaries c- divides d- reservoirs...
- C Chemistry How many protons are there in the neutral atom of 35(superscript)17(subscript)cl? a. 17 b. 19 c. 20 4. 35 5. 37 it would be great if you could tell me why, cause i really...
- C Chemistry When chemists write out reactions, they often place numbers to the left of a chemical formula. e.g. the first 2 in: 2h+o-- h2o. what do these numbers to the left of chemical...
- C Chemistry Which of the following reactions shows a chemical change producing a gas? apex...
- C Chemistry Water can be formed in the following reaction: 2h2+o2-- 2h2o if you have 10 moles of hydrogen gas (h2), how many moles of water can you form? (enter just the number: not...
- C Chemistry If you have 4.9 x 10^24 molecules of o2 at stp, how many liters of o2 do you have?...
- C Chemistry What is not a role of chemistry in modern agriculture...
- C Chemistry Compare and contrast adhesion and cohesion, using capillary action as an example....
- C Chemistry Very easy significant figures question! i m a dummy with significant figures, but my teacher is very strict on them my question here is what should this equation turn out...

Ответ:
T = 350 K
Explanation:
Given data:
Volume of balloon = 0.0733 L
Number of moles = 0.00230 mol
Pressure = 0.924 atm
Temperature in K = ?
Solution:
PV = nRT
PV / nR = T
T = 0.924 atm × 0.0733 L / 0.00230 mol × 0.0821 atm. L/mol. K
T = 0.07 / 0.0002 /K
T = 350 K