Robinlynn228
21.04.2020 •
Chemistry
Please help! use what you know of limiting reactants
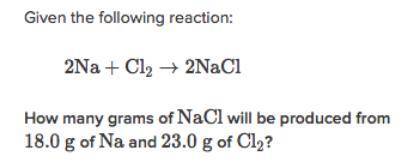
Solved
Show answers
More tips
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
- S Sport When and Where Will the 2014 World Cup be Held?...
Answers on questions: Chemistry
- M Mathematics Jane drew a triangle with all the angle being 70 degrees. The teacher explained to her that she was wrong. Explain why you think Jane is wrong....
- M Mathematics This equation has a positive slope true false...
- H History John locke believed there was a social contract between a government and its people. what were the details of this contract? a.) citizens should agree to obey their government...

Ответ:
Limiting reatant refers to a reatant that is used completely as the result its hinder the reaction to continue further .
In your problem you have to calculate number of moles for each reatant and the reatant having less moles is then limiting reatant . Then you will use it with its ratio to calculate t the moles of required . Then convert moles to grams
Ответ:
39.4 g
Explanation:
Of the 137.33 grams per mole of PCl₃, 106.36 grams of it are chlorine, about 77.4%. Thus the mass of chlorine in the 50.9 grams of PCl₃ will be ...
(50.9 g)(0.7745) ≈ 39.4 g
39.4 g of chlorine reacted with the available phosphorus to produce 50.9 g of phosphorus trichloride.
__
P = 30.974 g/mol
Cl = 35.453 g/mol
Comment on answer choices
Since our answer doesn't match any of the offered choices, we assume there is a typo in the question. The best choice of those shown here is 35.4 g. A little more than 3/4 of the product weight is chlorine, so the answers 66.4 and 17.7 or lower do not make any sense.