shelbyp2003
04.05.2021 •
Chemistry
PLEASE HELP, WILL GIVE BRAINLIEST
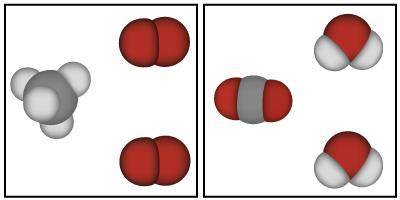
The image below shows models that represent the reactants and products of a chemical reaction.
Based on the image below, which of the following is true?
A. Mass could have either been gained or lost during this chemical reaction because atoms always change mass when they react.
B. Mass was lost in this chemical reaction because the atoms in the reactants are smaller than the atoms in the products.
C. Mass was conserved in this chemical reaction because the same atoms are present in both the products and the reactants.
D. Mass was gained in this chemical reaction because there are more atoms in the products than there were in the reactants.
Solved
Show answers
More tips
- S Science and Technology Exploring Our Galaxy: How Many Planets are in the Milky Way?...
- S Science and Technology Colliders: How They Work and Why They Matter...
- A Animals and plants Unraveling the Mystery of Loch Ness: What Does the Loch Ness Monster Look Like?...
- L Leisure and Entertainment How Many Seasons are There in the TV Show Interns?...
- S Sport Playing Bowling: Rules and Advice for Novices...
- L Leisure and Entertainment The Best Film of 2010: A Look Back at the Academy Awards...
- S Sport How to Learn Swimming? Simple Tips for Beginners...
- C Computers and Internet What is Web 2.0 and How Does it Work?...
- C Computers and Internet War of Social Media: Which Platform is the Leader?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
Answers on questions: Chemistry
- H History Why was germany s split between protestants in the north and catholics in the south important?...
- C Chemistry What does Delta S 0 mean in a reaction?...
- M Mathematics Choose the equation that could be used to find two consecutive integers whose sum is 67....
- C Chemistry The element magnesium would most likely have which one of these properties...

Ответ:
It would be d.
Step-by-step explanation:
:)