janahiac09
28.06.2019 •
Chemistry
Pls ! : ) based on valence electron configurations, which of the following is the accurate formula for a compound formed from sodium (na) and oxygen (: all numerical values below are subscripts) *
Solved
Show answers
More tips
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- L Leisure and Entertainment What to Bring on a Hike? Essential Items to Pack for a Safe and Enjoyable Adventure...
- L Leisure and Entertainment Couchsurfing: A New Way to Travel...
- S Style and Beauty Autotanning: Harmful or Safe?...
- F Food and Cooking 10 Ideas for a Wedding Anniversary Gift...
- H Health and Medicine How to Reduce Sweating in the Heat and Beyond: Say Goodbye to Excessive Sweat...
- F Food and Cooking Do Aphrodisiacs Really Work? Separating Fact from Fiction...
- H Health and Medicine What to Eat to Lose Weight?...
- A Animals and plants How to Teach Your Parrot to Talk?...
- H Health and Medicine How to Improve Your Posture?...
Answers on questions: Chemistry
- M Mathematics What are 3 things the number line can tell u? please help (TWT)...
- E English 6. Choose the best answer. Which of these sentences has only the linking verb italicized? please help it’s due in 45 minutes i appreciate it...
- M Mathematics Answer and explain ILL MARK BRAINLIEST...
- C Computers and Technology Free points If you wanna rp just tell me...
- M Mathematics Ihave 10 find each side length round to the nearest tenth if necessary...

Ответ:
The study of chemicals is called chemistry. There are two types of solution in the chemistry and these are acid and base.
The correct answer is
What are moles?
The mole is the base unit of the amount of substance in the International System of Units.It is defined as exactly 6.02214076×10²³ elementary entities, which may be atoms, molecules, ions, or electrons.Thus sodium needs to lose one electron in order to attain noble gas configuration and Oxygen needs to gain two electrons to attain noble gas configuration.
Thus Na will lose one electron to form and Oxygen gain two electrons to form
and Oxygen gain two electrons to form In order to form a stable neutral compound, two sodium ions are required.
In order to form a stable neutral compound, two sodium ions are required.
Thus the formula will be .
.
For more information, refer to the link:-
link
Ответ:
The accurate formula is .
.
Explanation: Atomic no of Sodium is 11. The electronic configuration is:
Na:
Atomic no of Oxygen is 8. The electronic configuration is:
O:
Thus sodium needs to lose one electron in order to attain noble gas configuration and Oxygen needs to gain two electrons to attain noble gas configuration.
Thus Na will lose one electron to form and Oxygen gain two electrons to form
and Oxygen gain two electrons to form 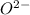 . In order to form a stable neutral compound, two sodium ions are required.
. In order to form a stable neutral compound, two sodium ions are required.
Thus the formula will be .
.
Ответ:
See explanation below
Explanation:
You are missing certain data. I found one, in the attached picture. All you have to do is replace your data in this procedure to get an accurate answer.
According to the picture attached, we have 2.4 g of HBr and 1.9 g of NaOH. This mix produces 0.411 g of water. To get the %yield of water, we first need to see how many theorical grams of water are formed. With this we can calculate the %yield.
The overall reaction is:
HBr + NaOH --------> NaBr + H₂O
We have a mole ratio of 1:1 so, let's see which is the limiting reactant between the acid and the base, using the reported molar mass for each (MM HBr = 80.9119 g/mol; MM NaOH = 39.997 g/mol)
moles HBr = 2.4 / 80.9119 = 0.02966 moles
moles NaOH = 1.9 / 39.997 = 0.0475 moles
So, we can clearly see that the moles of NaOH are in excess, so the HBr is the limiting reactant.
As we have a mole ratio of 1:1 with each compound here, and that the HBr is the limiting reactant we can say that:
moles HBr = moles H₂O = 0.02966 moles of water
Now, using the molar mass of water, let's calculate the theorical yield of water: (MM water = 18.01528 g/mol
m H₂O = 0.02966 * 18.01528 = 0.534 g
We have the theorical yield, let's calculate the % yield:
% = 0.411 / 0.534 * 100
% = 76.92%