Pls help it’s due in 6 minutes!!
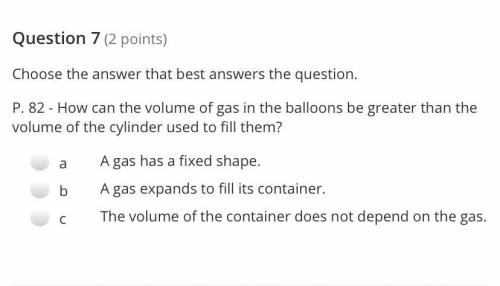
Solved
Show answers
More tips
- A Animals and plants Unraveling the Mystery of Loch Ness: What Does the Loch Ness Monster Look Like?...
- L Leisure and Entertainment Should You Buy a Ceramic Knife?...
- C Computers and Internet How to easily and quickly disable Firebug in Gmail and Google Docs...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Chemistry
- M Mathematics Based on the graph, what is the solution of the system of equatins?...
- E English In Who cares that he fell back to the sea? See him acclaiming the sun and come plunging down while his sensible daddy goes straight into town. , the word “sensible” is used...
- M Mathematics Which of the following is not true about intersecting lines...
- B Biology Which one of these is not an example of intercellular communication? question 5 options: a message travels between gap junctions. an electrical signal causes muscle cells...
- E English Which of the following can be reasonably inferred from the following two lines? Line #1: Miss Maudie’s hand touched mine and I answered mildly enough, “Nome, just a lady....

Ответ:
B✨
Explanation:
Ответ:
kindly, find the attached images.
Part 1: image 1.
Part 2: image 2.
Explanation:
Part 1: Describe how you can determine the total change in enthalpy and activation energy from the diagram, and if each is positive or negative.
Kindly find image 1.
The total change in enthalpy (ΔH) is the difference in potential energy between products and reactants. (P.E. of products > P.E. of reactants).ΔH = (P.E.) products - (P.E.) reactants.
So, it will be positive.
Activation energy is the difference between potential energy of the intermediate and reactants.It will be positive.
Part 2: Describe how the curve will look if the reaction was exothermic. Be sure to mention changes in the potential energies of the reactants and products and the sign changes of the enthalpy.
Kindly find image 2.
The total change in enthalpy (ΔH) is the difference in potential energy between products and reactants. (P.E. of products < P.E. of reactants).ΔH = (P.E.) products - (P.E.) reactants.
So, it will be neagtive.
Activation energy is the difference between potential energy of the intermediate and reactants.It will be positive.