QUESTÃO 1 - A equação que representa corretamente a combustão total do eteno, C2H4, é * A) C2H4 + O2 = 2 CO2 + 2 H2O B) C2H4 = 2 CO2 + 2 H2O C) C2H4 + 2 O2 = 2 CO2 + 2 H2O D) C2H4 + 3 O2 = 2 CO2 + 2 H2O E) C2H4 + 4 O2 = 2 CO2 + 4 H2O
Solved
Show answers
More tips
- C Computers and Internet How to Download Movies from Torrents?...
- F Food and Cooking How to Make the Perfect Glühwein: Step-by-Step Guide...
- A Animals and plants How to Grow Lime from a Seed: Simple Tips and Interesting Facts...
- S Style and Beauty How to Properly Tie a Tie: 5 Simple Steps...
- C Computers and Internet Dynamically Assigned IP Address: What Is It and How Does It Work?...
- C Computers and Internet How to Check the Speed of My Internet?...
- H Health and Medicine 5 Simple Steps to Quit Smoking for Good...
- C Computers and Internet How to Download Videos from YouTube? Simple Steps to Download Any Content...
- H Health and Medicine What is the Normal Blood Sugar Level in a Healthy Person?...
- S Style and Beauty How to Get Rid of Acne: Scientifically Proven Methods...
Answers on questions: Chemistry
- B Biology What is the external stimulus in thigmotropism?...
- B Biology When jackson throws a baseball in a straight path, what two forces cause the ball to eventually stop moving? a) gravity and friction b) mass and ground force c) wind resistance and...
- M Mathematics The random variables corresponding to the interarrival times of customers and the service times of the servers are commonly part of a(n) simulation....
- E English Which word or words should be capitalized in this sentence? we talked saturday afternoon about her strawberry-rhubarb pie entry in the festival. a. festival b. strawberry-rhubarb...

Ответ:
1)Thus based on the diagram , the values of principle quantum number that can be assigned to the electrons are : 2,3 and 4.
2)This quantum number tells us the shell in which an electron is present and how far it is roughly from the nucleus.
Explanation:
According to the rules of electron filling in subshells in quantum mechanics , Aufbau's principle states that :
"An electron must be filled in a lower energy state or subshell and then higher. If they have the same energy , the one with lesser principal quantum number will be filled first."
Thus ,
The series of filling goes like:
Thus , the subshells in the given information have 1,3 and 1 orbitals respectively. Thus it has to be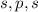 which is possible in two cases :
which is possible in two cases :
1)Thus based on the diagram , the values of principle quantum number that can be assigned to the electrons are : 2,3 and 4.
2)This quantum number tells us the shell in which an electron is present and how far it is roughly from the nucleus.