blakeesteigmanoxd9bd
27.12.2020 •
Chemistry
Show that the de Broglie wave length of an electron in the ground state of Hydrogen atom corresponds to the circumference of the circle having the Bohr radius as its radius.
Solved
Show answers
More tips
- L Leisure and Entertainment The Best Film of 2010: A Look Back at the Academy Awards...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- L Leisure and Entertainment What to Bring on a Hike? Essential Items to Pack for a Safe and Enjoyable Adventure...
- L Leisure and Entertainment Couchsurfing: A New Way to Travel...
- S Style and Beauty Autotanning: Harmful or Safe?...
- F Food and Cooking 10 Ideas for a Wedding Anniversary Gift...
- H Health and Medicine How to Reduce Sweating in the Heat and Beyond: Say Goodbye to Excessive Sweat...
- F Food and Cooking Do Aphrodisiacs Really Work? Separating Fact from Fiction...
- H Health and Medicine What to Eat to Lose Weight?...
- A Animals and plants How to Teach Your Parrot to Talk?...
Answers on questions: Chemistry
- M Mathematics What times what equals 30 but add to get 40?...
- B Business Long-term liability data for the budgeted balance sheet is derived from...
- S Social Studies When andrew sees his grandmother placing curlers in her hair, he tries to imitate her. his grandmother says, no, no! that is not the way boys get ready to go. she takes...
- S Social Studies The famous “2½ ton truck” was used to bring food for these 73,000 U.S. soldiers to the depot. How many of these trucks should be loaded per day just to carry food?...

Ответ:
2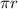 = nλ
= nλ
Explanation:
Without mincing words let's dive straight into the solution to the above question. From the question above, we are given the need to start by using the de Broglie wavelength, this can be represented mathematically by the equation below:
h/ mv = λ. --------------------------------------------------------------------------------------------[1]. Where λ is the de Broglie wave length.
Starting by writing the mathematical representation for the angular momentum as given below:
Thus, 2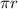 = n[h/mv] ----------------------------------------------------------------------------[2].
= n[h/mv] ----------------------------------------------------------------------------[2].
Recall from equation [1] that h/ mv = λ. Therefore,
2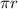 = nλ. --------------------------------------------------------------------------------[3].
= nλ. --------------------------------------------------------------------------------[3].
The equation above, that is equation [3] has proved that de Broglie wave length of an electron in the ground state of Hydrogen atom corresponds to the circumference of the circle having the Bohr radius as its radius.
Ответ:
D,C
Explanation: