ariannecama
16.03.2020 •
Chemistry
Small quantities of oxygen can be prepared in the laboratory by heating potassium chlorate, KClO 3 ( s ) . The equation for the reaction is 2 KClO 3 ⟶ 2 KCl + 3 O 2 Calculate how many grams of O 2 ( g ) can be produced from heating 82.4 g KClO 3 ( s ) .
Solved
Show answers
More tips
- C Computers and Internet Why is Yandex s robot Bless.yandex.ru Important?...
- S Science and Technology How to Restore the Diamond Shine to Your Tarnished Silverware...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
- L Leisure and Entertainment How to Find a Phone Number by Address: The Ultimate Guide...
- F Food and Cooking Pu-erh Tea: History, Varieties, Benefits, and Risks...
- S Style and Beauty Why is Sugaring Better than Waxing for Hair Removal?...
- C Computers and Internet How to Delete a Page on VKontakte: The Ultimate Guide from Experts...
- F Food and Cooking How to Make Delicious Plov: Secrets and Recipes...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
- L Leisure and Entertainment Unlocking the Secrets of Winning the Lottery...
Answers on questions: Chemistry
- C Chemistry A. what happens in the nucleus of an atom when an alpha particle is emitted? b. what happens in the nucleus of an atom when a beta particle is emitted?...
- C Chemistry Particles with negative charges are called positively charged particles are called particles with no charge are called...
- C Chemistry An air mass usually forms in an area of low pressure because it s stable low pressure because it s unstable high pressure because it s stable high pressure because it s unstable...
- C Chemistry What term describes the rise in prices of most goods and services?...
- C Chemistry 2) Does the atomic number of an atom change if it is an ion? Why or why not?...
- C Chemistry training doctors to recognize child abuse and explain how you could apply it to teaching. Write a short paragraph, using complete sentences with proper grammar, spelling, and...
- C Chemistry Which element is always found in impure form a. copperb. nitrogenc. hydrogenkindly answer as fast as possible : )...
- C Chemistry What is the average speed of a bird that flies 1.55 km in 30 minutes...
- M Mathematics Which is 1/3 + 1/4? Plz help...
- H History What did France do in order to solve the slave uprising in Haiti?...

Ответ:
Mass of produced = 32 g
produced = 32 g
Explanation:
Calculation of the moles of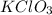 as:-
as:-
Mass = 82.4 g
Molar mass of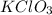 = 122.55 g/mol
= 122.55 g/mol
The formula for the calculation of moles is shown below:
Thus,
From the reaction shown below:-
2 moles of potassium chlorate on reaction forms 3 moles of oxygen gas
So,
0.67237 moles of potassium chlorate on reaction forms moles of oxygen gas
moles of oxygen gas
Moles of oxygen gas = 1 mole
Molar mass of oxygen gas = 32 g/mol
Mass of produced = 32 g
produced = 32 g
Ответ: