geezelouise808
26.05.2021 •
Chemistry
So, i know that if a metal mixes with an acid it produces salt + hydrogen
and i was just wondering... what happens if a METAL OXIDE is mixed with an acid ?
thanksss, <3
Solved
Show answers
More tips
- F Family and Home How to Choose the Perfect Air Conditioner for Your Life...
- H Health and Medicine Discover the Hidden Principles and Real Results of the Japanese Diet...
- H Health and Medicine Understanding Pregnancy Tests: What You Need to Know?...
- H Health and Medicine What Makes a Man a Man?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
Answers on questions: Chemistry
- C Chemistry Which statement best represents a proper hypothesis? I think there is no effect on hearing if you listen to loud music for only three hours a day. Water will flow...
- C Chemistry True or false? scientists who study processes that take millions of years cannot conduct experiments...
- C Chemistry 2. Name 3 elements that are found in the same period. 3. Identify each of the following a. Metal: I b. Nonmetal: c. Metalloid: d. Alkali Metal: e Halogen: f Noble...
- C Chemistry Que significa HI en educación ambiental...
- C Chemistry What happens to the electron configuration when you move across a period to the right?...
- C Chemistry If noble gases dont react in most conditions how is xe4f4 is formed...
- C Chemistry The breakdown of glycogen (an energy-storage compound) is an example of a(n) reaction....
- C Chemistry Asample of ammonia has a mass of 43.5 g. how many molecules are in this sample?...
- C Chemistry Do noble gases react with other elements and why...
- C Chemistry Are noble gases the only stable elements in periodic table and why...

Ответ:
The evidence that can demonstrate the covalent nature of water is
is
Further Explanation:
The bond that is formed by the mutual sharing of electrons between the bonded atoms is called a covalent bond. It is also known as a molecular bond. Covalent compounds are those compounds which are formed by the electron sharing between two or more non-metals.
Following are some of the properties of covalent compounds:
1. These are brittle solids.
2. Low melting and boiling points.
3. Poor conductors of heat and electricity.
4. Low enthalpy of fusion.
Boron trichloride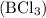 , silicon dioxide
, silicon dioxide  , and xenon hexafluoride
, and xenon hexafluoride  are some examples of covalent compounds.
are some examples of covalent compounds.
(A). Water is a liquid that has a definite volume. The physical property can be determined without knowing the chemical nature of the substance. Therefore the liquid state of water with a fixed volume is not able to predict its chemical properties and determination of the type of compound comes under these properties. So it cannot predict the covalent nature of water. Hence, the option (A) is incorrect.
(B). Boiling of water on a kitchen stove does not allow its temperature to reach a certain higher limit. This indicates that it boils at a moderate temperature due to which it is clear that water has low boiling point. As we know, boiling points of covalent compounds are low due to the presence of weak intermolecular forces between them. This proves water is a covalent compound. Hence, the option (B) is correct
(C). Water is a covalent compound. As we know covalent compounds are formed by the mutual sharing of electrons. So their electrical conductivity of covalent compounds is very low due to the absence of free ions in them and therefore this property cannot show the covalent nature of water molecule. Hence, the option (C) is incorrect
(D). Water can change its shape and can be sprayed or poured. This shows water has some fluidity in it and has intermediate forces of attraction. But it gives no idea about whether water is a covalent compound or not. So this property cannot be used to predict the covalent nature of water molecule. Hence, the option (D) is incorrect
Learn more:
1. Identification of ionic bonding:
2. Example of physical change:
Answer details:
Grade: High School
Subject: Chemistry
Chapter: Ionic and covalent compounds
Keywords: water, covalent compound, H2O, mutual sharing, electrons, liquid, shape, volume, kitchen stove, electrical conductivity, covalent bond, molecular bond.