Ezonthekid
09.07.2019 •
Chemistry
Sodium hydroxide reacts with carbon dioxide to form sodium carbonate and water: 2 naoh(s) + co2(g) → na2co3(s) + h2o(l) how many grams of na2co3 can be prepared from 2.40 g of naoh?
Solved
Show answers
More tips
- F Family and Home 5 Tips for Choosing Toys for Your Child...
- L Leisure and Entertainment How to Find a Phone Number by Address: The Ultimate Guide...
- P Philosophy How to Properly Create a Vision Board?...
- C Computers and Internet What is Web 2.0 and How Does it Work?...
- S Style and Beauty Is Photoepilation the Solution to Unwanted Hair Forever?...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...

Ответ:
Answer : The mass of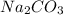 prepared can be 3.18 grams.
prepared can be 3.18 grams.
Explanation : Given,
Mass of NaOH = 2.40 g
Molar mass of NaOH = 40 g/mole
Molar mass of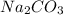 = 106 g/mole
= 106 g/mole
First we have to calculate the moles of .
.
Now we have to calculate the moles of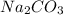 .
.
The balanced chemical reaction is,
From the balanced reaction we conclude that,
As, 2 moles of react to give 1 mole of
react to give 1 mole of 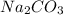
So, 0.06 moles of react to give
react to give 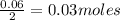 of
of 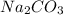
Now we have to calculate the mass of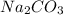 .
.
Therefore, the mass of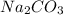 prepared can be 3.18 grams.
prepared can be 3.18 grams.
Ответ: