thefootballman1115
05.11.2020 •
Chemistry
Someone has left a shiny metal bowl outside in the sun. Which may have the potential to damage your eyes: looking at the outside of the bowl or the inside of the bowl? Explain.
Solved
Show answers
More tips
- W Work and Career What is the Most In-Demand Profession in the Modern World?...
- A Auto and Moto How Can Parking Sensors Help Drivers?...
- H Health and Medicine What is the Normal Blood Sugar Level in a Healthy Person?...
- F Food and Cooking Red Caviar: How to Choose the Best?...
- C Computers and Internet Clearing Cache: How to Speed Up Your Browser...
- S Style and Beauty How are artificial nails removed?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
- F Food and Cooking Everything You Need to Know About Pasta...
Answers on questions: Chemistry
- C Chemistry What is true when a reaction has reached equilibrium? the concentration of products and reactants is constant. the reaction has stopped. the concentration of the products...
- H History What Constitutional provision protects most civil liberties for US citizens?...
- M Mathematics Decide whether 2 x + 8 and 2(× + 4) + 4 are equivalent expression. explain how you know....
- E English Dally said cherry s friend, Marsha, was acting as a spy for the Greasers? 1. True 2. False...

Ответ:
Looking at the inside of the bowl
Explanation:
The curvature of the bowl would likely focus the light to a single point or area above it. This higher intensity area would be more dangerous to look at.
Ответ:
Ответ:
The empirical and molecular formula for the given organic compound is CH and and it is not an alkane.
and it is not an alkane.
Explanation:
The chemical equation for the combustion of hydrocarbon having carbon and hydrogen follows:
where, 'x' and 'y' are the subscripts of Carbon, hydrogen and oxygen respectively.
We are given:
Mass of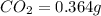
Mass of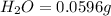
We know that:
Molar mass of carbon dioxide = 44 g/mol
Molar mass of water = 18 g/mol
For calculating the mass of carbon:
In 44 g of carbon dioxide, 12 g of carbon is contained.
So, in 0.364 g of carbon dioxide, of carbon will be contained.
of carbon will be contained.
For calculating the mass of hydrogen:
In 18 g of water, 2 g of hydrogen is contained.
So, in 0.0596 g of water,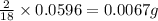 of hydrogen will be contained.
of hydrogen will be contained.
To formulate the empirical formula, we need to follow some steps:
Step 1: Converting the given masses into moles.Moles of Carbon =
Moles of Hydrogen =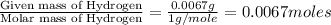 ]
]
Step 2: Calculating the mole ratio of the given elements.For the mole ratio, we divide each value of the moles by the smallest number of moles calculated which is 0.0067 moles.
For Carbon =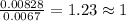
For Hydrogen =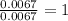
Step 3: Taking the mole ratio as their subscripts.The ratio of C : H = 1 : 1
The empirical formula for the given compound is CH
For determining the molecular formula, we need to determine the valency which is multiplied by each element to get the molecular formula.
The equation used to calculate the valency is:
We are given:
Mass of molecular formula = 128.2 g/mol
Mass of empirical formula = 13 g/mol
Putting values in above equation, we get:
Multiplying this valency by the subscript of every element of empirical formula, we get:
The general formula of an alkane is , where n = any natural number
, where n = any natural number
Here, n = 10 and it does not satisfy being an alkane
Hence, the empirical and molecular formula for the given organic compound is CH and and it is not an alkane.
and it is not an alkane.