Space vehicles use solid lithium hydroxide to remove exhaled carbon dioxide according to the equation: 2LiOH + CO 2 Li 2 CO 3 + H 2 O. Determine the mass of carbon dioxide removed if the space vehicle carries 42.0 mol of LiOH.
Solved
Show answers
More tips
- T Travel and tourism How to Use a Compass: A Beginner s Guide...
- C Computers and Internet Porn Banner: What It Is and How to Get Rid Of It?...
- F Food and Cooking How many stages of coffee roasting are there?...
- F Food and Cooking From Latte to Espresso: Which Coffee Drink is the Most Popular on Earth?...
- F Food and Cooking Experts Name Top 5 Healthiest Teas...
- C Computers and Internet Google Search Tips and Tricks: Everything You Need to Know...
- H Health and Medicine Discover the Hidden Principles and Real Results of the Japanese Diet...
- H Health and Medicine Liver Cleansing - Rejuvenation for the Body?...
- S Style and Beauty Is Photoepilation the Solution to Unwanted Hair Forever?...
- S Style and Beauty Unbelievable Hairstyles for Long Hair - 2021 Trends...
Answers on questions: Chemistry
- C Chemistry in two or more complete sentences, describe the five step process of fractional distillation that is used to refine crude oil....
- C Chemistry Acetylene, C2H2, can be converted to ethane, C2H6, by a process known as hydrogenation. The reaction is: C2H2 + 2H2 ⇄ C2H6 The Kp at standard conditions for the reaction...
- C Chemistry How many molecules are there in 0.655 moles of C6H14...
- C Chemistry When reacting Na with Cl2 , we calculated that the theoretical yield should be 12.5 grams. Our actual yield was 13.0 grams. What is the percent yield?...
- C Chemistry Zn + 2HCl → ZnCl2 + H2 15 grams of HCl should theoretically produce 0.42 grams of H2 . The reaction actually produced 0.15 grams of H2 . What is the percent yield...
- C Chemistry All elements in Group 18 are called and are , which means they don’t readily react with other elements....
- C Chemistry Kilogram The volume of helium in a blimp is 6.28 x 10°milliliters. The density of helium in the blimp is 0.1786 meter3 Find the mass of the helium in the blimp. (Hint:...
- C Chemistry During what stage of the cell cycle does replication occur...
- C Chemistry Identify the precipitate (if any) that forms when koh and cu(no3)2 are mixed...
- C Chemistry The name of the metal in an ionic compounds does not change true or false...

Ответ:
924g
Explanation:
Given parameters
Number of moles of LiOH = 42moles
Unknown:
Mass of carbon dioxide removed = ?
Solution:
We have to solve this problem from the known specie to the unknown. Since we are dealing with reactants, the compound with given number of moles is the known specie.
Using the balanced chemical reaction, we can deduce the number of moles of that of the unknown;
2LiOH + CO₂ → Li₂CO₃ + H₂O
we see that;
2 moles of LiOH will remove 1 mole of CO₂
42 moles of LiOH will remove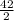 = 21moles of CO₂
= 21moles of CO₂
We know the number of moles of CO₂ removed, let us find the mass;
Mass of CO₂ = number of moles x molar mass
Molar mass of CO₂ = 12 + 2(32) = 44g/mol
Mass of CO₂ = 21 x 44 = 924g
Ответ: