StupidFatChipmunk
16.04.2021 •
Chemistry
Stoichiometry chem problem
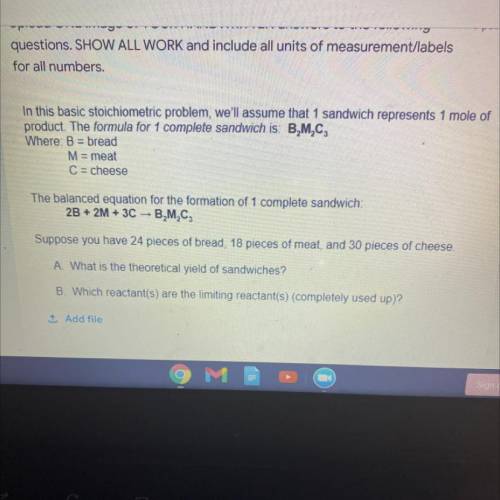
Solved
Show answers
More tips
- F Food and Cooking Why Doesn t the Confirmation Link Come to Email?...
- L Leisure and Entertainment What Movies You Should Watch: A Guideline to Make the Right Decision...
- F Family and Home How to Choose a Baby Stroller: Tips from Experienced Parents...
- H Health and Medicine 5 Simple Steps to Quit Smoking for Good...
- H Health and Medicine Coughing: Causes, Types, and Treatment Methods...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
Answers on questions: Chemistry
- C Chemistry 20 Which of these statements about plants are true? 1 plants use calcium to make chlorophyll 2 plants are the producers in a food chain 3 plants are herbivores 4 plants...
- M Mathematics Kendall ordered a set of blue and yellow pins. She received 30 pins in all. 6 of the pins were blue. What percentage of the pins were blue?...
- S Social Studies Critics of president polk and his decision to go to war included winfield scott zachary taylor abraham lincoln robert e. lee...
- B Business State and explain ways to set-up business in domestic market?...
- H History Has anyone taken the 8th grade final exam on segment two part 1 of history in the wisconsin virtual school? it covers module 5 and 6. looking for on what to study....
- E English Why does Katniss cut the branch during the anthem? A. The loud noise will mask the sound of her sawing away at the branch B. As an act of rebellion C. To honor the...

Ответ:
the solute here is NH₃ because it's the one with less amount. which makes water the solvent.
1) let's convert the grams of NH₃ to moles using the molar mass
molar mass of NH₃= 14.0 + (3 x 1.01)= 17.03 g/ mol
30.0 g (1 mol/ 17.03 g)= 1.76 mol NH₃
2) let's convert the grams of water into kilograms (just divide by 1000)
70.0g= 0.0700 kg
3) let's plug in the values into the molality formula
molality= mol/ Kg---> 1.76 mol/ 0.070 kg= 25.1 m