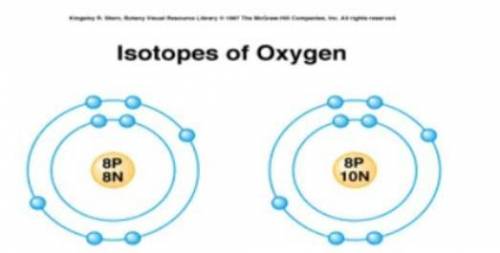
Students were shown models of two atoms and asked to make a list of similarities and differences between the models.
Which of the statements about the atomic models shown is correct?
A) Both models represent atoms with the same atomic number.
B) Both models represent atoms with the same atomic mass.
C) The model on the left is an ion and the model on the right is an isotope.
D) The model on the left is an isotope and the model on the right is an ion.
Solved
Show answers
More tips
- H Health and Medicine Sunstroke: Causes, Symptoms, and Precautions...
- F Family and Home Do You Really Need a Playpen for Your Child?...
- F Food and Cooking Do Aphrodisiacs Really Work? Separating Fact from Fiction...
- C Computers and Internet Log-in: what is it and why do you need it?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Chemistry
- M Mathematics The population, P(t), in millions, of a country, in year t, is given by the formula P(t) = 24 + 0.4t. What are the values of the population for t = 10, 20, and 30?...
- B Business Genting Berhad is a Malaysian conglomerate with holdings in plantations and tourist resorts. The beta estimated for the firm, relative to the Malaysian stock exchange is...
- M Mathematics Write an expression that represents the area of the rectangle. Remember A = LW a)6x2−3x b)9x2−3x c)6x-1 d)12x - 2...

Ответ:
A: Both models represent atoms with the same atomic number.
Explanation:
The atomic number equals the number of protons.
Ответ:
stop being lazy and do youre own work lol
Step-by-step explanation: