Suppose of hcl and of o2 are added to an empty flask. how much will be in the flask at equilibrium? a. none. b. some, but less than 155. mmol, c. 155. mmol, d. more than 155. mmol
Solved
Show answers
More tips
- F Family and Home How to Choose a Baby Stroller: Tips from Experienced Parents...
- H Health and Medicine 5 Simple Steps to Quit Smoking for Good...
- C Computers and Internet How to Download Videos from YouTube? Simple Steps to Download Any Content...
- H Health and Medicine What is the Normal Blood Sugar Level in a Healthy Person?...
- S Style and Beauty How to Get Rid of Acne: Scientifically Proven Methods...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- C Computers and Internet How to Delete Your Account on Odnoklassniki...
- H Health and Medicine 10 Simple and Effective Tips on How to Lose Weight in a Week...
- F Food and Cooking What s the Best Rice for Cooking Plov?...
- F Family and Home How to Get Rid of Your Neighbors?...
Answers on questions: Chemistry
- C Chemistry 1.) what are the two broad categories that elements can be divided into? 2.) list the names of the two elements and three compounds found in air....
- C Chemistry which is the correctly balanced equation for the reaction of rust, fe2o3, and hydrochloric acid, hcl?...
- C Chemistry What is the molar mass of a gas if 18.0 L of the gas at STP has a mass of 41.6 grams? O 9.69 g O 33.4 g O 1.86 g O 51.8 g...
- C Chemistry 50 g of NaF is added to 100 mL of water to form a solution. What is the concentration of the solution?...
- C Chemistry The process that is responsible for the continuous removal of carbon dioxide from the atmosphere is:...
- C Chemistry Asmall cube of aluminum measures15.6mm on a side and weights 10.25g. what is the density of aluminum in g/cm3...
- C Chemistry Predict how decreasing the volume of the container used for the reaction below would affect the initial rate of the reaction, and explain your prediction.. h2 (g)...
- C Chemistry Calculate the hydronium ion concentration in an aqueous solution that contains 2.50 × 10-2 mol L-1 in hydroxide ion....
- C Chemistry Based on this information what is the most likely reason for refrigerating most foods reduce the rate at which they spoil...
- B Business I like becuase u get to learn...

Ответ:
This is an incomplete question, here is a complete question.
Hydrogen chloride and oxygen react to form water and chlorine, like this:
Use this chemical equation to answer the questions in the table below.
Suppose 155. mmol of HCl and 38.8 mmol of O₂ are added to an empty flask. How much HCl will be in the flask at equilibrium?
(1) None.
(2) Some, but less than 155. mmol,
(3) 155. mmol,
(4) More than 155. mmol,
Answer : The correct option is, (1) None
Explanation :
The given balanced chemical reaction is:
From the balanced chemical reaction, we conclude that
As, 4 mmole of HCl react with 1 mmoles of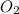
So, 155. mmole of HCl react with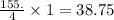 mmoles of
mmoles of 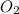
That means,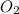 is excess reagent and
is excess reagent and 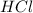 is a limiting reagent. All the moles of HCl will consume.
is a limiting reagent. All the moles of HCl will consume.
Hence, the correct option is, None.
Ответ:
Atoms consist of a central, dense nucleus that is surrounded by one or more lightweight negatively charged particles called electrons. The nucleus is made up of positively charged particles called protons and neutrons which are neutral.