maxy7347go
07.10.2019 •
Chemistry
Suppose that you are climbing a high mountain and the oxygen partial pressure in the air is reduced to 75 torr. estimate the percentage of the oxygen carrying capacity that will be utilized, assuming that the ph of both tissues and lungs is 7.4 and that the oxygen concentration in the tissues is 20 torr. show all work. i need to know how to work this problem out. the answer is 62.7%. show all work and i will award 5 stars.
Solved
Show answers
More tips
- F Family and Home Do Lullabies Help Babies Sleep or Is it Just a Myth?...
- H Health and Medicine Tick Traps: How to Remove Them Safely and Effectively...
- S Society and Politics Why are thugs called gopniks ? A fascinating journey through Russian subculture...
- A Animals and plants Want a Perfect Lawn? Learn How to Plant Grass the Right Way...
- A Animals and plants How to Properly Care for a Pet Decorative Rabbit at Home?...
- C Computers and Internet How to Check the Speed of My Internet?...
- H Health and Medicine 10 Ways to Cleanse Your Colon and Improve Your Health...
- W Work and Career How to Write a Resume That Catches the Employer s Attention?...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...

Ответ:
Explanation:
The given data is as follows.
Initial atmospheric pressure of the O = 760 torr
The partial pressure of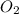 =
= 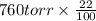
= 167.2 torr
Partial pressure of oxygen (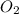 ) in the blood is 20 torr.
) in the blood is 20 torr.
Therefore, calculate the carrying capacity as follows.
Carrying capacity =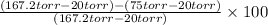
=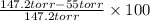
= 0.6263
or, = 62.6%
Thus, we can conclude that the oxygen carrying capacity is 62.6%.
Ответ:
big boys
red chair
please make me Brainliest