henriquetucker
26.06.2020 •
Chemistry
Suppose you are working with a NaOH stock solution but you need a solution with a lower concentration for your experiment. Calculate the volume of the 1.475 M stock NaOH solution needed to prepare 250.0 mL of 0.1374 M dilute NaOH solution.
Solved
Show answers
More tips
- H Health and Medicine Get Rid of Warts: Simple and Effective Ways...
- S Style and Beauty How to Choose the Perfect Hair Color?...
- C Computers and Internet Best iPad Games: Our Opinion...
- A Animals and plants Man s Best Friend: Which Dog Breed Is the Most Friendly?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
- G Goods and services How to Choose the Right High Chair for Your Baby?...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
- S Style and Beauty How to braid friendship bracelets?...
Answers on questions: Chemistry
- C Chemistry Will mark as brainliest. explain why can two nonmetals bond together, but two metals cannot? a) metals will only form covalent bonds. b) nonmetals can share pairs of electrons and...
- E English ( Jabari unmasked) Why does the speaker feel compelled to hide their identity or present it in a certain way?...
- B Business Choice of ownership determines the degree to which each owner has personal liability for the firm s debts and the sources of funds available to the firm to finance future expansion....
- H History 1. How did physical geography affect the development of Indus Valley civilizations? 2. What physical feature separates India from the continent of Asia? 3. What positive and negative...
- M Mathematics The equation y = 0.75x + 1.5 models the data on a scientist’s scatter plot, where x is the amount of water a plant receives in milliliters, and y is the height in centimeters. if...
- P Physics Which of the following is an example of erosion A• to wear down and carry away rocks and soil through the force of gravity B• to wear down and carry away rock and soil through the...

Ответ:
Explanation:
In this case, we have a dilution problem. We have to remember that in the dilution procedure we go from a solution with higher concentration to a solution with lesser concentration. Therefore we have to start with the dilution equation:
Now we can identify the variables:
If we plug all the values into the equation:
And we solve for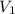 :
:
I hope it helps!
Ответ: