Suppose you had a hypothesis you believed to be correct. what should you do to make it become a theory? a. write a letter to the nobel prize committee. b. do the same experiment over and over again. c. get all your friends to say that they agree with you. d. collect more evidence and try to convince other scientists.
Solved
Show answers
More tips
- S Style and Beauty Is Photoepilation the Solution to Unwanted Hair Forever?...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
Answers on questions: Chemistry
- E English 8. read these lines from alfred, lord tennyson’s “the lady of shalott.” “on either side the river lie/long fields of barley and of rye,/that clothe the wold and meet the sky;/and...
- M Mathematics Give an example of reciprocals?...
- M Mathematics Simplify and place the rounded answer on the top line...
- M Mathematics PLS HELP ME WITH THIS GEOMETRY QUESTION...

Ответ:
I believe the answer is D. Incredibly sorry if I am wrong, but it seems that to make it a theory you should collect evidence to show to other scientists.
Ответ:
I believe the answer is D as well
Ответ:
The molar mass of the protein is 13392.86 g/mol
Explanation:
To calculate the concentration of solute, we use the equation for osmotic pressure, which is:
where,
i = Van't hoff factor = 1 (for non-electrolytes)
M = molarity of solute = ?
R = Gas constant =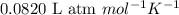
T = temperature of the solution =![28^oC=[273+28]=301K](/tpl/images/0229/0989/3c69a.png)
Putting values in above equation, we get:
To calculate the molecular mass of solute, we use the equation used to calculate the molarity of solution:
We are given:
Molarity of solution = 0.0056 M
Given mass of protein = 0.150 g
Volume of solution = 2 mL
Putting values in above equation, we get:
Hence, the molar mass of the protein is 13392.86 g/mol