donahueno1753
14.12.2020 •
Chemistry
Tara's lab group was investigating changes in matter. They measured the properties of a liquid
sample, such as its melting point, density, and boiling point. They then passed an electric current
through the liquid and observed the formation of bubbles from each of the electrical contacts. The
remaining liquid appeared unchanged and had the same properties as before the electric current
was applied. When the gases were tested, each gas had a different set of properties.
Based on these observations, what can Tara's group infer about the original liquid?
A. It was a colloid.
B. It was a pure substance.
c. It was a solution.
D. It was a suspension.
Solved
Show answers
More tips
- G Goods and services Which TV is better - LCD or Plasma?...
- S Sport How to Learn to Pull Up on Monkey Bars?...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- C Computers and Internet Where did torrents.ru move to?...
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
- G Goods and services How to Choose the Right High Chair for Your Baby?...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
Answers on questions: Chemistry
- M Mathematics Find f(g(4)). f(g(4)) =...
- M Mathematics Lin’s needs 192 cupcakes each baking sheet can make 16 cupcakes how many sheets are needed to make all cupcakes...
- M Mathematics SOMEONE PLEASE HELP ME WITH THIS ASAP....
- M Mathematics The weather service measures the temperature, T, every hour, h, beginning at midnight of each 24-hour period. On Friday, the temperature was modeled by T= h +8. Which is represented...

Ответ:
pOH of resulting solution is 0.086
Explanation:
KOH and CsOH are monoacidic strong base
Number of moles of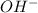 in 375 mL of 0.88 M of KOH =
in 375 mL of 0.88 M of KOH = 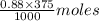 = 0.33 moles
= 0.33 moles
Number of moles of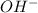 in 496 mL of 0.76 M of CsOH =
in 496 mL of 0.76 M of CsOH = 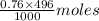 = 0.38 moles
= 0.38 moles
Total volume of mixture = (375 + 496) mL = 871 mL
Total number of moles of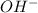 in mixture = (0.33 + 0.38) moles = 0.71 moles
in mixture = (0.33 + 0.38) moles = 0.71 moles
So, concentration of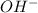 in mixture,
in mixture, ![[OH^{-}]](/tpl/images/0538/8908/e46dd.png) =
= 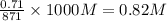
Hence,![pOH=-log[OH^{-}]=-log(0.82)=0.086](/tpl/images/0538/8908/1f1db.png)