maryam2863
04.12.2019 •
Chemistry
The acid-dissociation constants of hc3h5o3 and ch3nh3+ are given in the table below. which of the following mixtures is a buffer with a ph of approximately 3?
hc3h5o3
ch3nh3+
ka
8.3 x 10-4
2.3 x 10-11
a mixture of 100. ml of 0.1 m nac3h5o3 and 100. ml of naoh
a mixture of 100. ml of 0.1 m ch3nh3cl and 100. ml of ch3nh2
a mixture of 100. ml of 0.1 m hc3h5o3 and 50. ml of naoh
a mixture of 100. ml of 0.1 m ch3nh3cl and 50. ml of naoh
Solved
Show answers
More tips
- A Auto and Moto Which alarm system to choose?...
- A Auto and Moto How to Start a Diesel Engine in Cold Weather?...
- H Health and Medicine Hangover: How to Get Rid of It Quickly?...
- C Computers and Internet How to Calibrate Your Monitor: Useful Tips and Recommendations...
- P Philosophy Agnosticism: Opinion or Belief?...
- L Leisure and Entertainment When will Maslenitsa start?...
- F Family and Home Stay Warm but Don t Overheat: What is the Optimal Temperature for Your Home During Winter?...
- H Health and Medicine Heartburn: Causes and Ways to Get Rid of It...
- S Society and Politics Will Japan become Russia s Military Enemy?...
- F Family and Home How to Choose the Perfect Air Conditioner for Your Life...
Answers on questions: Chemistry
- C Chemistry A student measures the depth of as swimming pool to be 2.04 m at its deepest end. The accepted value is 2 m. What is his % error?...
- H Health How can you boost your self-esteem?...
- B Biology Eight components of Nucleic Acids are listed in the box. Which components bond with adenine in a section of double-stranded DNA? 3, 4, 7, and 8 only 2 and 4 only 3 and 5 only 1,...
- A Arts Which technique or process did Auguste Rodin use in this sculpture? the subtractive technique of carving in this in-the-round sculpture the subtractive technique of carving in...

Ответ:
A mixture of 100. mL of 0.1 M HC3H5O3 and 50. mL of NaOH
Explanation:
The pH of a buffer solution is calculated using following relation
Thus the pH of buffer solution will be near to the pKa of the acid used in making the buffer solution.
The pKa value of HC₃H₅O₃ acid is more closer to required pH = 4 than CH₃NH₃⁺ acid.
pKa = -log [Ka]
For HC₃H₅O₃
pKa = 3.1
For CH₃NH₃⁺
pKa = 10.64
pKb = 14-10.64 = 3.36 [Thus the pKb of this acid is also near to required pH value)
A mixture of 100. mL of 0.1 M HC3H5O3 and 50. mL of NaOH
Half of the acid will get neutralized by the given base and thus will result in equal concentration of both the weak acid and the salt making the pH just equal to the pKa value.
Ответ:
The branch of science which deals with the chemicals and the bond is called chemistry.
The correct answer is C.
The solution in which the ph remains constant is called buffer solution.
The equation used to solve the question is as follows:-
Thus the pH of the buffer solution will be near to the pKa of the acid used in making the buffer solution because the salt is used in the question with respect to the acid.
The pKa value of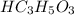 acid is closer to the required pH = 4 than
acid is closer to the required pH = 4 than 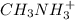 acid.
acid.
The total pKb value is
Hence, the correct answer is C that is A mixture of 100 mL of 0.1 M HC3H5O3 and 50. mL of NaOH
For more information, refer to the link:-
Ответ:
Water is polar molecule, ionic compound dissolves in it as like dissolves like.
there is ionic interaction between a salt and water
Sugar has hydroxyl group in it. These hydroxyl group are able to form hydrogen bond with water molecule.
[Hydrogen bond : the interaction between a hydrogen, bonded with a highly electro-negative element (F,O,N) and other highly electro-negative element (F,O,N)]
Thus due to hydrogen bond sugar is soluble in water.
Explanation:The water molecules, , are polar, with positive partial charges on the hydrogen and a negative partial charge on the oxygen. This makes it possible to interact with ionic compounds such as salt (NaCl). This interaction occurs through the partially positive and negative ends of the water, so that the positive charges attract the negative ones. The NaCl salt in water dissociates (separates) into and ions. ions are surrounded by negative partial charges of water oxygen, while ions are surrounded by hydrogen ends with positive partial charge. In this way the salt is dissolved in water.
Sugar is a molecular compound formed by covalent bonds. In a polar covalent bond, electrons are shared unevenly. This creates a slightly negatively charged end and a slightly positively charged end. This is what happens with sugar. These extremes are those that interact with the positive and negative extremes of water, mentioned previously. Then it is possible to say that sugar dissolves in water because both substances are polar substances.
In short, water dissolves most of the substances that are polar or ionic, as in the case of sugar and salt.