The atoms in barium metal are arranged in a bodycentered cubic unit cell. Calculate the radius of a barium atom if the density of barium is 3.50 g?cm23 . Hint: Use your answer to Exercise 4.18.
Solved
Show answers
More tips
- W Work and Career Secrets of a Super Supervisor: Proper Team Management...
- P Photography and Videography Understanding HDR: How It Works and Why You Need It...
- G Goods and services Which TV is better - LCD or Plasma?...
- S Sport How to Learn to Pull Up on Monkey Bars?...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- C Computers and Internet Where did torrents.ru move to?...
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
- G Goods and services How to Choose the Right High Chair for Your Baby?...
Answers on questions: Chemistry
- C Chemistry Which of the following measurement tools has greater measurement uncertainty: a meter stick that measures to the nearest centimeter (1 cm = 0.01 m) or a ruler that measures...
- C Chemistry Draw molecular structure of MgCl2...
- C Chemistry From the following data calculate the potassium content per tablet in effervescent Kch bicarbonate tablets. Weight of 20 tablets = 35.6751 g Weight of tablet powder taken for...
- C Chemistry A mixture has no fixed composition, whereas a compound has” Explain what this means, using hydrogen, oxygen, and water to illustrate your answer....
- C Chemistry From 1940, there have been four main laboratories that have worked to produce elements with atomic numbers greater than 92. Describe what IUPAC’s role would be in such a situation....
- C Chemistry PL HEL How is Endoplasmic Reticulum alike to a factory?...
- C Chemistry Lake Okeechobee, a major agricultural area, water conservation areas, and the Everglades wetlands are projects maintained by a group that works with NASA to help Florida. Which...
- C Chemistry Significant figures in the measurement 0.070020 meter?...
- C Chemistry What amount of copper is deposited when 13.0g of zinc reacts with excess copper (ll) tetraoxosulphate (IV) solution according to the following equation? Zn (s) + CuSO4(aq) →...
- C Chemistry If the stoichiometric ratio of your reaction were 1:1 what volumes would you use to collect four data points on each side of the highest temperature? what if it were 3:1? 4:1?...

Ответ:
The radius of a barium atom is r = 2.19 10^-8
10^-8
Explanation:
The atomic weight of barium is 137.34.
The body-centered cubic structure has two atoms per unit cell.
Therefore, the mass of Ba in a unit cell is calculated as,
volume = mass / density
volume =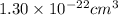
The edge length of a cube then is the cube root of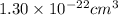 or
or
The body diagonal is 4 x the radius and equals a 1.732,
Therefore r = (a 1.732) / 4
1.732) / 4
= (5.06 10^-8
10^-8
The radius of a barium atom is cm.
cm.
Ответ:
A. 1
Explanation:
The higher you get in the atmosphere the cooler the temperature will be. The reason it's actually colder is because, as you go up in the atmosphere, the Earth's atmosphere feels less pressure the higher up you go. So as the gas in the atmosphere rises it feels less pressure, which makes it expand.
Hope this Help :)
Please mark brainliest :D