ged32802p4gf0r
20.02.2020 •
Chemistry
The azimuthal quantum number for the outermost electrons in a nitrogen atom in the ground state is .
Solved
Show answers
More tips
- H Health and Medicine Mercury Thermometer Danger: What to do when a thermometer breaks?...
- F Food and Cooking How to cook crayfish? Everything you need to know...
- G Goods and services LED-подсветка в LCD-телевизорах: 5 причин, почему она лучше других технологий...
- P Photography and Videography Understanding HDR: How It Works and Why You Need It...
- G Goods and services Which TV is better - LCD or Plasma?...
- S Sport How to Learn to Pull Up on Monkey Bars?...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- C Computers and Internet Where did torrents.ru move to?...
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
Answers on questions: Chemistry
- M Mathematics Find the measurement of angle X A) 91° C) 1360 B) 1050 D) 100°...
- H History How many hispanics have lived in the southwest since this territory was part of mexico?...
- E English Which story detail supports Malu s statement This isn t home ? A. Malu does not try to help her mom unlock the door B. Malu will not step on the mat in front of the door...
- M Mathematics Can someone that actually has a brain help me, please. I will give out brainliest. please don t steal these points I really need help....

Ответ:
Quantum number are the numbers assigned to the electrons that represents its energy, position, and spin. The azimuthal quantum number for the outermost electrons in a nitrogen atom in the ground state is 1.
Quantum Number:
These are the number assigned to the electrons, that represents its energy, position, and spin.
There are 4 quantum number,
Principal Quantum number(n)Azimuthal quantum number(Azimuthal quantum number(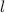 ):
):
This quantum number show the orbital structure. This quantum number represented by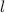 . It can be calculated by the formula (n-1).
. It can be calculated by the formula (n-1).
For s-orbital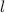 = 0
= 0
For p-orbital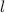 = 1
= 1
For d-orbital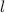 = 2
= 2
Since, the outermost electron of the nitrogen atom occur in the p-orbital.
Therefore, the azimuthal quantum number for the outermost electrons in a nitrogen atom in the ground state is 1.
To know more about quantum numbers, refer to the link:
link
Ответ:
1
Explanation:
The azimuthal quantum number shows the energy sublevels in each energy levels. It ranges from 0-n-1. The outermost electrons of nitrogen are in 2p orbital. The p orbital has an azimuthal quantum number of 1.
Ответ: